(a) Draw the structure of the product that is formed when the following compound is heated with...
Question:
(a) Draw the structure of the product that is formed when the following compound is heated with one equivalent of sodium methoxide in methanol. Explain your reasoning.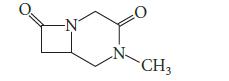
(b) What would the product be if the same compound were heated with a large excess of aqueous H2SO4?
Transcribed Image Text:
-N N 0 CH3
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 80% (5 reviews)
a There are two cyclic amide or lactam functional g...View the full answer

Answered By

Ashington Waweru
I am a lecturer, research writer and also a qualified financial analyst and accountant. I am qualified and articulate in many disciplines including English, Accounting, Finance, Quantitative spreadsheet analysis, Economics, and Statistics. I am an expert with sixteen years of experience in online industry-related work. I have a master's in business administration and a bachelor’s degree in education, accounting, and economics options.
I am a writer and proofreading expert with sixteen years of experience in online writing, proofreading, and text editing. I have vast knowledge and experience in writing techniques and styles such as APA, ASA, MLA, Chicago, Turabian, IEEE, and many others.
I am also an online blogger and research writer with sixteen years of writing and proofreading articles and reports. I have written many scripts and articles for blogs, and I also specialize in search engine
I have sixteen years of experience in Excel data entry, Excel data analysis, R-studio quantitative analysis, SPSS quantitative analysis, research writing, and proofreading articles and reports. I will deliver the highest quality online and offline Excel, R, SPSS, and other spreadsheet solutions within your operational deadlines. I have also compiled many original Excel quantitative and text spreadsheets which solve client’s problems in my research writing career.
I have extensive enterprise resource planning accounting, financial modeling, financial reporting, and company analysis: customer relationship management, enterprise resource planning, financial accounting projects, and corporate finance.
I am articulate in psychology, engineering, nursing, counseling, project management, accounting, finance, quantitative spreadsheet analysis, statistical and economic analysis, among many other industry fields and academic disciplines. I work to solve problems and provide accurate and credible solutions and research reports in all industries in the global economy.
I have taught and conducted masters and Ph.D. thesis research for specialists in Quantitative finance, Financial Accounting, Actuarial science, Macroeconomics, Microeconomics, Risk Management, Managerial Economics, Engineering Economics, Financial economics, Taxation and many other disciplines including water engineering, psychology, e-commerce, mechanical engineering, leadership and many others.
I have developed many courses on online websites like Teachable and Thinkific. I also developed an accounting reporting automation software project for Utafiti sacco located at ILRI Uthiru Kenya when I was working there in year 2001.
I am a mature, self-motivated worker who delivers high-quality, on-time reports which solve client’s problems accurately.
I have written many academic and professional industry research papers and tutored many clients from college to university undergraduate, master's and Ph.D. students, and corporate professionals. I anticipate your hiring me.
I know I will deliver the highest quality work you will find anywhere to award me your project work. Please note that I am looking for a long-term work relationship with you. I look forward to you delivering the best service to you.
3.00+
2+ Reviews
10+ Question Solved
Related Book For 

Question Posted:
Students also viewed these Sciences questions
-
1. Write a java program for the following:- Make an array containing a few Strings. Sort it by Length (i.e., shortest to longest) Reverse length (i.e., longest to shortest) Alphabetically by the...
-
Write a program to pass two numbers from main function to function foo, swap the numbers in a function foo and print the swapped value in main function using call by value method.
-
Draw the structure of the product with molecular formula C 10 H 10 O that is obtained when the compound below is heated with aqueous acid. CN CN C10H100 Heat
-
Suppose that the vice president of marketing asks you to write a program to create labels for a onetime advertising promotion. As IT manager, you know that the labels can be prepared more efficiently...
-
Alto Publishers Companys selected accounts as of November 30, 2012, follow: Requirements 1. Prepare the multi-step income statement, statement of retained earnings, and balance sheet for the first...
-
What is the difference between pure arbitrage and risk arbitrage? If an investor observes the price of a stock trading in one exchange to be different from its price in another exchange, what form of...
-
Copy your worksheet from Question 6 into another worksheet. Change the increase from 10% to 18%. Protect the worksheet, so that changes cannot be made. Question 6 Open a new spreadsheet. Type...
-
Perdon Corporation manufactures safes'large mobile safes, and large walkin stationary bank safes. As part of its annual budgeting process, Perdon is analyzing the profitability of its two products....
-
1. Solve the double inequality below. Then graph the solution set on the real number line. -81+3(x-2) < 13
-
Complete the following reactions by giving the principal organic product(s). (b) (c) Raney Ni (catalyst) heat PhCHC=N + H O II EtO-C-CH-CN LiAlH4 (excess) 1) H3O+ 2) "OH 0-C-CH3 T Ph-CH-COEt + LiAlH4...
-
Give the structure of the product in the reaction of each of the following esters with isotopically labeled sodium hydroxide, Na + 18 OH and explain your reasoning. PhCH-O- A -CH3 PhCH-0-C-CH3 B
-
At what temperature will nitrogen gas (N2) have the same rms speed as helium (He) when the helium is at 20.0C?
-
Should You Lay Off Employees in Need? Jorge Pine is a restauranteur who attributes success to three elements: great food, great operations, and enough capital to support them. However, after opening...
-
What is the role of a claims handler and loss adjuster in the claims process? Explain
-
Why is a tax allowance important to understand?
-
Who sells a stock warrant?
-
What is quality for you? Give an example of brand of product or service that conforms to your definition. Explain your reason briefly.
-
Refer to the income statement given in the preceding exercise. Prepare a new contribution income statement for the Nantucket Inn in each of the following independent situations. (Ignore income...
-
H Corporation has a bond outstanding. It has a coupon rate of 8 percent and a $1000 par value. The bond has 6 years left to maturity but could be called after three years for $1000 plus a call...
-
For each of the following alcohols, write a retrosynthetic analysis and synthesis that involves an appropriate organometallic reagent (either a Grignard or alkyllithium reagent). (a) (b) (c) OH
-
Synthesize each of the following compounds starting from primary or secondary alcohols containing seven carbons or less and, if appropriate, bromobenzene. (a) (b) (c)
-
The alcohol shown here is used in making perfumes. Write a retrosynthetic analysis and then synthetic reactions that could be used to prepare this alcohol from bromobenzene and 1-butene.
-
nx (a) Let f (0, 1) R be given by fn(x) = +1. Show that {f} converges pointwise to a continuous function f, but the convergence is not uniform. (Remark: This shows that pointwise convergence to a...
-
Problem 5 (3 points each) Let and g be bounded functions on [a, b]. (a) Prove the triangle inequality for the uniform norm, || + 9||u ||f||u+9u (b) Using your result in (a), prove the reverse...
-
A particular mathematics textbook measures 8.5 inches by 11 inches by 1.5 inches. How many of these textbooks can be placed into a shipping box that measures 12 inches by 12 inches by 6 inches? How...

Study smarter with the SolutionInn App


