A peptide was subjected to one cycle of the Edman degradation, and the following compound was obtained.
Question:
A peptide was subjected to one cycle of the Edman degradation, and the following compound was obtained. What is the amino-terminal residue of the peptide?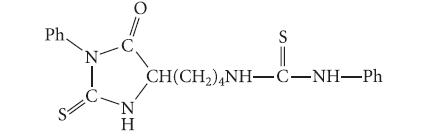
Transcribed Image Text:
Ph. S= N -C ZH O N Η S CH(CH,)NH–C-NH—Ph
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 100% (2 reviews)
The aminoterminal residue is lysine Both the sidech...View the full answer

Answered By

Akash Goel
I am in the teaching field since 2008 when i was enrolled myself in chartered accountants course
Since then i have an experience of teaching of class XI, XII, BCOM, MCOM, MBA, CA CPT.
5.00+
1+ Reviews
10+ Question Solved
Related Book For 

Question Posted:
Students also viewed these Sciences questions
-
A peptide C was found to have a molecular mass of about 1000. Amino acid analysis of C revealed its composition to be (Ala 2 ,Arg,Gly,Ile). The peptide was unchanged on treatment with the Edman...
-
The Sanger method for N-terminus determination is a less common alternative to the Edman degradation. In the Sanger method, the peptide is treated with the Sanger reagent, 2, 4-dinitrofluorobenzene,...
-
Proteins are synthesized with a particular amino acid sequence through the translation of information encoded in messenger RNA by an RNAprotein complex called a ribosome. Amino acids are specified by...
-
Tort cases are so common that it is likely you or someone you know has been involved in a tort case. If so, share what the case was about, what the outcome was, and how you felt about the case and...
-
Distinguish between a. Capital expenditures and revenue expenditures. How is each accounted for? b. Ordinary repairs and improvements. How is each accounted for?
-
Carbohydrates Anonymous (CA) operates a chain of weight-loss centers for carb lovers. Its services have been in great demand in recent years and its profits have soared. CA recently paid an annual...
-
An investment is guaranteed to have a unique value of IRR if which of the following is true? a. Alternating positive and negative cash flows b. An initial negative cash flow followed by all positive...
-
Preston Village engaged in the following transactions: 1. It issued $ 20 million in bonds to purchase a new municipal ofce building. The proceeds were recorded in a capital projects fund. 2. It...
-
In one month Ashton earned $800 at her part-time job, and $80 was withheld for federal income tax. Suppose she earns $1200 next month. How much will be withheld for federal income tax?
-
A peptide Q has the following composition by amino acid analysis: Treatment of Q once with the Edman reagent followed by anhydrous acid gives a new peptide R with the following composition by amino...
-
Which of the following statements would correctly describe the isoelectric point of cysteic acid, an oxidation product of cysteine? Explain your answer. (1) Lower than that of aspartic acid (2) About...
-
The major determinant of a countrys income distribution is its distribution of productive and income-earning assets. Explain the meaning of this statement, giving examples of different kinds of...
-
For this week, read the following assigned articles: - Delk, C. W. (2019). Leadership: A Simple Situational Approach: Strategic Leadership for a practical leadership style. Leadership Excellence,...
-
Departmentalisation plays an influential role in achieving organisational objectives. Provide a critical evaluation on the various forms of departmentalisation in business.
-
What are the costs of implementing an accounting change? What are some other possible costs of changing accounting methods? Can you think of examples other than the ones provided in your reading this...
-
Karen is trying to price a 2-year annual coupon bond with a coupon rate of 8%, and a par value of $1000. The current price of 1-year and 2-year $1000 STRIPS are $950 and $870, respectively. What is...
-
Given the following code, what is the life of each variable listed; public class Widgets; { { } private int numberOfWidgets; public Widgets(int number) } { numberOfWidgets = number; public void...
-
Alex has just graduated from college and has accepted a job at a different city. Rather than move his furniture, Alex decides to sell it. Alex sets his price by noting that removing the bolts that...
-
Making use of the tables of atomic masses, find the velocity with which the products of the reaction B10 (n, ) Li7 come apart; the reaction proceeds via interaction of very slow neutrons with...
-
The standard free energy of activation (G++) for hydration of 2-methylpropene to 2-methyl-2-propanol (Eq. 4.41, p. 169) is 91.3 kJ mol-t (Zt 8 kcal mol-l;. The standard free energy G for hydration of...
-
Iodine azide,I-Nr, adds to isobutylene in the following manner: iodine azide
-
For each of the following cases, provide the structure of an alkene that would give the alcohol as the major (or only) product of hydroboration--oxidation. CH CH CH CH OH CH, CH,
-
Part A- Describe how to make supply meet demand over the medium term? Part B - How difficult is it to forecast demand over a 3 to 5 year period? Part C - Look up online the typical costs to hire and...
-
Deckko Co owns two firms: a dairy company and a snack company. The dairy company has stable, predictable sales. The snack company is constantly innovating to try and discover a high-value product,...
-
Who carries out the role of process improvement and why? What is the role of a Leader in the Coaching Kata mentality

Study smarter with the SolutionInn App


