(a) Predict the stereochemistry of compounds B and C in Fig. P28.31. (b) What stereoisomer of A...
Question:
(a) Predict the stereochemistry of compounds B and C in Fig. P28.31.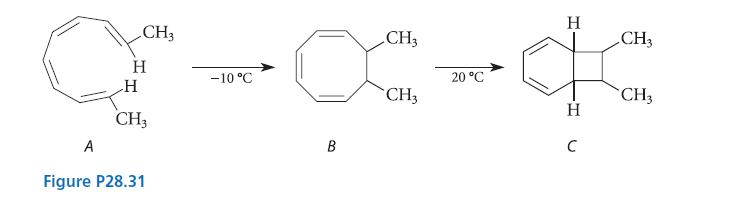
(b) What stereoisomer of A also gives compound C on heating?
Transcribed Image Text:
A CH3 H H CH3 Figure P28.31 -10 °C H CH3 ·x-da 20 °C CH3 B H C CH3 CH3
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 0% (2 reviews)
a b Because it involves 4n electrons and thermal conditions the ...View the full answer

Answered By

Jeff Omollo
As an educator I have had the opportunity to work with students of all ages and backgrounds. Throughout my career, I have developed a teaching style that encourages student engagement and promotes active learning. My education and tutoring skills has enabled me to empower students to become lifelong learners.
5.00+
5+ Reviews
42+ Question Solved
Related Book For 

Question Posted:
Students also viewed these Sciences questions
-
In 1985, two researchers at the University of California, Riverside, carried out the reaction given in Fig. P28.51. The equilibrium mixture contained compound A (22%), a single stereoisomer of B...
-
In 1985, two researchers at the University of California, Riverside, carried out the reaction given in Fig. P27.51. The equilibrium mixture contained compound A (22%), a single stereoisomer of B...
-
Compound A has two stereoisomers, but compounds B and C exist as single compounds. Explain. CH CH CH CH2 HCH-CH2 CH2CH3 CH3
-
Evaluate the limit or state that it does not exist. lim (x,y) (1,-3) In(3x + y)
-
Notes on Net Assets. The following items are taken from the financial statements of the Kids Clubs of America for the years ending December 31, 2011, and 2010, with related notes. Required a. Explain...
-
Bellati Inc. produces large industrial machinery. Bellati has a machining department and a group of direct laborers called machinists. Each machinist is paid $50,000 and can machine up to 500 units...
-
X rays with a wavelength of \(0.12 \mathrm{~nm}\) undergo first-order diffraction from a crystal at a \(68^{\circ}\) angle of incidence. What is the angle of second-order diffraction?
-
Transactions involving the common stock account of Higrade Gas Company during the 2-year period 2011 to 2012 were as follows: 2011 Jan. 1 Had a balance of 200,000 shares of $10 par common stock ....
-
Find the amplitude, phase shift, and period of the function. (2)+3 y=-2 sin 2x Give the exact values, not decimal approximations. Amplitude: Phase shift: Period: JT
-
Show by an orbital symmetry analysis that a [3,3] sigmatropic reaction that is antarafacial on both components is allowed. Would you expect such a reaction to be very common? Why?
-
(a) Give the curved-arrow mechanism for the anionic oxyCope reaction of compound A, and explain why the stereoisomer B does not react under the same conditions. (b) Give the structure of the product,...
-
Ram holds a $400,000 note receivable from Pat. It has been learned that Pat filed for Chapter 7 bankruptcy and that the expected recovery of nonsecured claims is 45 on the dollar. Inventory items...
-
Describe the current tax law for sale of residence.
-
Does the Internal Revenue Code of 1986 include pre-1986 tax law?
-
Chris is starting a new business called Church Company. What entity structure may he use?
-
Explain and differentiate between the three concepts of income: economic, legal, and accounting.
-
What is the difference between a current distribution and a liquidating distribution?
-
Compute r2 for Problem 12.24 (Problem 12.6). Discuss the value of r2 obtained. (in Problem12.6) x 12 2 28 17 15 22 19 24
-
After Theorem 1.5 we note that multiplying a row by 0 is not allowed because that could change a solution set. Give an example of a system with solution set S0 where after multiplying a row by 0 the...
-
Give the substitutive name for each of the following compounds. (a) CH3CH2CH2CH2OH (b) (c) (d) HO CI H,C CH3 H3C-C-CH3 SH
-
The effectiveness of barbiturates as sedatives has been found to be directly related to their solubility in, and thus their ability to penetrate, the lipid bilayers of membranes. Which of the...
-
A student, Flick Flaskflinger, in his twelfth year of graduate work, needed to prepare ethylmagnesium brdmide from ethyl bromide and magnesium, but found that his laboratory was out of diethyl ether....
-
1. Propose ONE new product or service that you should produce or develop for a new startup business through e-commerce web application. Give reasons and justify why you suggest that product. 2....
-
Summarize the steps associated with a decision-making process. You may answer using a written paragraph or diagram.
-
Do you think performance management is future orientated? substantiate your answer by referring your answer by referring to specific methods of performance appraisal.

Study smarter with the SolutionInn App


