In 1985, two researchers at the University of California, Riverside, carried out the reaction given in Fig.
Question:
In 1985, two researchers at the University of California, Riverside, carried out the reaction given in Fig. P28.51.
The equilibrium mixture contained compound A (22%), a single stereoisomer of B (47%), and a single stereoisomer of C (31%). Predict the stereochemistry of compounds B and C at the carbon marked with the asterisk (*). Explain your prediction.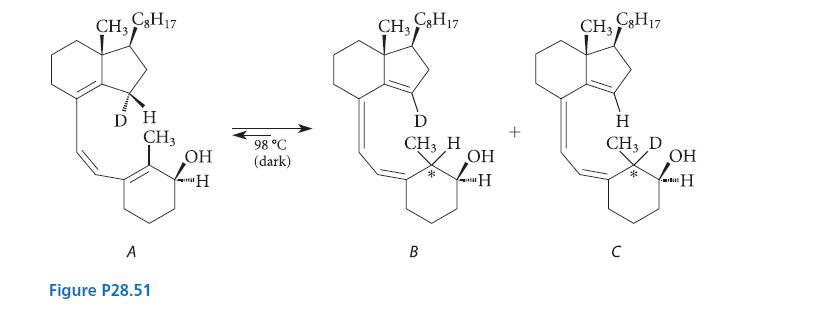
Transcribed Image Text:
CH₂ C3H17 DH A CH3 Figure P28.51 OH 'H 98 °C (dark) CH₂ C8H17 D CH3 H B * OH "H t CH₂ C8H17 H CH3 D C * OH H
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 100% (1 review)
This is a 17 sigmatropic rearrangement which according to ...View the full answer

Answered By

Rinki Devi
Professional, Experienced, and Expert tutor who will provide speedy and to-the-point solutions.
Hi there! Are you looking for a committed, reliable, and enthusiastic tutor? Well, teaching and learning are more of a second nature to me, having been raised by parents who are both teachers. I have done plenty of studying and lots of learning on many exciting and challenging topics. All these experiences have influenced my decision to take on the teaching role in various capacities. As a tutor, I am looking forward to getting to understand your needs and helping you achieve your academic goals. I'm highly flexible and contactable. I am available to work on short notice since I only prefer to work with very small and select groups of students.
I have been teaching students for 5 years now in different subjects and it's truly been one of the most rewarding experiences of my life. I have also done one-to-one tutoring with 100+ students and helped them achieve great subject knowledge.
5.00+
2+ Reviews
10+ Question Solved
Related Book For 

Question Posted:
Students also viewed these Sciences questions
-
In 1985, two researchers at the University of California, Riverside, carried out the reaction given in Fig. P27.51. The equilibrium mixture contained compound A (22%), a single stereoisomer of B...
-
Read the case study and answer the question below with a one page response. What does a SWOT analysis reveal about the overall attractiveness of Under Armours situation? Founded in 1996 by former...
-
Researchers at the University of Utah carried out a study to see if the size of the fork used to eat dinner has an effect on how much food is consumed (Food Network Magazine, January 2012). The...
-
At fiscal year-end December 31, 2015, Shop-World had the following assets and liabilities on its balance sheet (in millions): Current liabilities ............ $9,459 Long-term debt .................
-
At the beginning of the year, Plummer's Sports Center bought three used fitness machines from Advantage, Inc. The machines immediately were overhauled, installed, and started operating. The machines...
-
The Jenkins Corporation, a merchandising company, wants to include the cost of operating its ware-house in the cost of inventory. The companys controller argues that the cost of the warehouse is a...
-
Mrs. Clarks Foods was an Iowa company engaged in the business of distributing juice beverages. International Suntrade and Miller & Smith Foods were Canadian companies that acted as brokers...
-
Support-department cost allocation: direct and step-down methods. Phoenix Partners provides management consulting services to government and corporate clients. Phoenix has two support...
-
Assume a simultaneous open market purchase of 100 million from the Bank of England and a repayment of a discount loan of 5 million from Bank A to the Bank of England. Show the overall change in their...
-
An interesting heterocyclic compound C was prepared and trapped by the sequence of reactions given in Fig. P28.52. Give the structure of all missing compounds, and explain what happens in each...
-
Ions as well as neutral molecules undergo pericyclic reactions. Classify the pericyclic reactions of the cation involved in the trans formation shown in Fig. P28.49. Tell whether the methyl groups...
-
Given the following joint probability distribution of returns for securities A and B, calculate the covariance between the two securities. State Securiey A SecuriyB B Probability 10% 12 .10 .25 35...
-
Consider the below code. #include using namespace std; class Count { public: int count; }; Count(int c) { } } count = c; Count() { count- 0; void increment (Count c, int times) c.count++; times++; }...
-
A 0.40-kg mass is attached to a spring with a force constant of k = 257 N/m, and the mass-spring system is set into oscillation with an amplitude of A = 3.5 cm. Determine the following. (a)...
-
Week 5 Section B: Create a comprehensive system for human talent management (W5LO1, W5LO2. W5L03, CLO2, CLO3, and CLO 6) In this task, you will demonstrate your ability to develop a comprehensive...
-
. An inverted pendulum is supported by a linear spring, as shown in Fig. 1.76. It is required to: (a) Derive the equation for the angular motion 6(r) about O. (b) Determine the equilibrium positions.
-
3. Suppose we have a sorted array of 5000 elements. What is the maximum number of comparisons done by the Binary Search algorithm? Show work. 4. Give the Big O notation for the following functions...
-
Assume a firm is deciding between labor hours and machine hours as a cost driver. When would these drivers yield the same allocations?
-
Match each of the key terms with the definition that best fits it. _______________ A record of the sequence of data entries and the date of those entries. Here are the key terms from the chapter. The...
-
Bradykinin is a nonapeptide released by blood plasma globulins in response to a wasp sting. It is a very potent pain-causing agent. Its constituent amino acids are 2R, G, 2F, 3P, S. The use of 2,...
-
Complete hydrolysis of a heptapeptide showed that it has the following constituent amino acids: 2A, E, L, K, F, V Deduce the amino acid sequence of this heptapeptide from the following data. 1....
-
Part of the evidence for restricted rotation about the carbon-nitrogen bond in a peptide linkage comes from 1H NMR studies done with simple amides. For example, at room temperature the 1H NMR...
-
Fill in the missing amounts in each of the eight case situations below. Each case is independent of the others. (Hint: One way to find the missing amounts would be to prepare a contribution format...
-
Lucido Products markets two computer games: Claimjumper and Makeover. A contribution format income statement for a recent month for the two games appears below: Sales Variable expenses Claimjumper $...
-
Summer Company sells all its output at 25 percent above cost. Parade Corporation purchases all its inventory from Summer. Selected information on the operations of the companies over the past three...

Study smarter with the SolutionInn App


