Account with a mechanism for the fact that the hydrolysis of trimethyl phosphate to dimethyl phosphate in
Question:
Account with a mechanism for the fact that the hydrolysis of trimethyl phosphate to dimethyl phosphate in acidic solution containing 18O-labeled water gives methanol containing 18O and dimethyl phosphate containing no isotope, as shown in Fig. P25.30.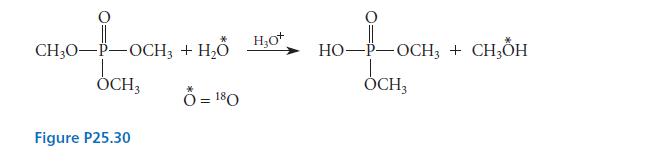
Transcribed Image Text:
40-100 CH₂0-P-OCH3 OCH3 Figure P25.30 + H₂O H₂O * Ô =180 H₂0+ 10-1-OCK HO-P-OCH3 + CH3OH OCH3
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 100% (1 review)
CH0P OCH3 HO ...View the full answer

Answered By

Asim farooq
I have done MS finance and expertise in the field of Accounting, finance, cost accounting, security analysis and portfolio management and management, MS office is at my fingertips, I want my client to take advantage of my practical knowledge. I have been mentoring my client on a freelancer website from last two years, Currently I am working in Telecom company as a financial analyst and before that working as an accountant with Pepsi for one year. I also join a nonprofit organization as a finance assistant to my job duties are making payment to client after tax calculation, I have started my professional career from teaching I was teaching to a master's level student for two years in the evening.
My Expert Service
Financial accounting, Financial management, Cost accounting, Human resource management, Business communication and report writing. Financial accounting : • Journal entries • Financial statements including balance sheet, Profit & Loss account, Cash flow statement • Adjustment entries • Ratio analysis • Accounting concepts • Single entry accounting • Double entry accounting • Bills of exchange • Bank reconciliation statements Cost accounting : • Budgeting • Job order costing • Process costing • Cost of goods sold Financial management : • Capital budgeting • Net Present Value (NPV) • Internal Rate of Return (IRR) • Payback period • Discounted cash flows • Financial analysis • Capital assets pricing model • Simple interest, Compound interest & annuities
4.40+
65+ Reviews
86+ Question Solved
Related Book For 

Question Posted:
Students also viewed these Sciences questions
-
The mechanism for acidic hydrolysis of a nitrile resembles the basic hydrolysis, except that the nitrile is first protonated, activating it toward attack by a weak nucleophile (water). Under acidic...
-
Suppose we have some optically pure (R)-2-butyl acetate that has been "labeled" with the heavy 18O isotope at one oxygen atom as shown. (a) Draw a mechanism for the hydrolysis of this compound under...
-
The pesticide Atrazine (C 8 H 14 C1N 5 , mol wt. 216 g/ mol) degrades in soil by a first-order reaction process. Consider the situation shown in the figure below, where there is a spill of solid...
-
Mick Stone disposed of the following assets during tax year 2020-21: (1) On 19 May 2020, Mick sold a freehold warehouse for 522,000. This warehouse was purchased on 6 August 2008 for 258,000, and was...
-
A 0.05394-kg sample of 144 Nd (atomic mass 143.91 u) emits an average of 2.36 particles each second. Find the decay constant in s 1 and the half-life in years.
-
Jan and Deana have been dreaming about owning a boat for some time and have decided that estimating its cash flows will help them in their decision process. They expect to have a disposable annual...
-
Explain stereotype threat effects and provide some examples of how they might occur in organizations and the consequences. What can organizations do to prevent stereotype threat effects?
-
Consider the following three mutually exclusive alternatives: Assuming that Alternatives B and C are replaced with: identical replacements at the end of their useful lives. and an 8% interest rate,...
-
How does data management within an information system impact the effectiveness and success of laboratory projects?
-
The reaction of water with metaphosphate ion is shown in Eq. 25.19b. Could nitrate ion undergo an analogous reaction with aqueous NaOH? If so, draw the structure of the product. If not, explain why....
-
Methanol containing the oxygen isotope 18 O is allowed to react, in separate reactions, with each of the acid chlorides shown in Fig. P25.29ac, and each of the resulting compounds A, C, and E is...
-
On January 2, 20X2, EL Limited established a subsidiary in Mexico City, Mexico. The subsidiary was named GC Company and the cost of ELs investment was C$ 500,000. When this investment was translated...
-
Alexandria currently has $80,000 in wealth saved up from her private speech therapy practice. Alexandria plans on working for 15 more years and is afraid theres a 5% chance that she will face a...
-
Describe three economic or social changes that have contributed to the trends in labor force participation rates for men and women since the 1950s. How have these changes impacted the trade-offs...
-
a. In 2018, the United States imposed tariffs of 20% to 50% on imported home washing machines. Explain how these tariffs affect the price that Whirlpool (a U.S. manufacturer) charges for washing...
-
What does it mean if a macroeconomic variable is a leading indicator? A lagging indicator? Give some examples of both leading and lagging indicators.
-
Youre a project manager overseeing five teams that are developing a new app. Each team must complete their work by July 1 in order to release the app by the end of the year. Based on your work...
-
The average cost of a one-bedroom apartment in a town is $550 per month. What is the probability of randomly selecting a sample of 50 one-bedroom apartments in this town and getting a sample mean of...
-
If (x) 0 on the interval [a, b], the definite integral gives the exact area under the curve between x = a and x = b.
-
Identify each of the following compounds from their spectra. (a) Compound A: molecular mass 113; gives a positive hydroxamate test; IR 2237, 1733, 1200 cm-1; proton NMR: 1.33 (3H, t, J = 7 Hz), ...
-
Outline a synthesis of each of the following compounds from the indicated starting materials and any other reagents. (a) 1-cyclohexyl-2-methyl-2-prupanol from bromocyclohexane (b) PhNHCH2CH2CH(CH3)2...
-
Rationalize each of the reactions in Fig. P21.56 with a mechanism, using the curved-arrow notation where possible. In part (d), identify compound A and show the mechanism for its formation. (Do not...
-
Louis is 25 years old and has a dream of having $1,000,000 in his investment account by the time he turns 50. If he invests in a fund that returns 5% per year (and compounds daily), how much does he...
-
Question 1 One year from today, investors anticipate that Amazing Inc. stock will pay a dividend of $3.25 per share. After that, investors believe that the dividend will grow at 20% per year for...
-
For the following problems, name the type of probability distribution that can be used for each case. 1. Of all customers purchasing automatic garage door openers, 75% purchase a chain-driven model....

Study smarter with the SolutionInn App


