Arrange the following alcohols according to increasing rates of their acid-catalyzed dehydration to alkene (smallest rate first),
Question:
Arrange the following alcohols according to increasing rates of their acid-catalyzed dehydration to alkene (smallest rate first), and explain your reasoning.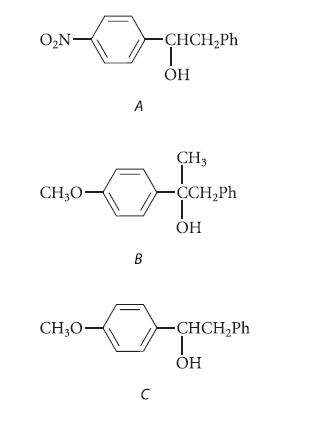
Transcribed Image Text:
O₂N- CH₂0- CH₂0- A B C -CHCH₂Ph T OH CH3 T -CCH₂Ph T OH -CHCH₂Ph T OH
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 100% (QA)
Acidcatalyzed dehydration involves a carbocation intermediate The pro...View the full answer

Answered By

Aquib jaleel Mooppan M A
After completing my bachelors in Mechanical engineering i was tutoring other engineering students and also xii students to excel in their exams i was also taking ielts class in order to apply for masters degree in Germany.
I tutored mostly college students and elementary school students privately after school and during the summer. We met in their homes or at the public library. I provided any necessary materials. I never had to advertise — word of mouth was how most folks knew of me. I stayed very busy, especially in the summers, and always had a full schedule. I typically met with each student's teacher in order to get an idea of what the focus of my instruction/remediation should be. Becoming familiar with the student's learning style(s) was also very helpful. I thoroughly enjoy teaching a class in a school situation.
My students and I had fun, and grew quite fond of one another.I had to take a break from tutoring due to academic constrictions and i miss my students very much.
0.00
0 Reviews
10+ Question Solved
Related Book For 

Question Posted:
Students also viewed these Sciences questions
-
Arrange the following alcohols in order of their reactivity toward acid-catalyzed dehydration (with the most reactive first): 1-Pentanol 2-Methyl-2-butanol 3-Methyl-2-butanol
-
Within series, arrange the compounds according to increasing rates of their reactions by the SN1 - E1 mechanism. Explain your reasoning. CH CH Br
-
Within each series, arrange the compounds according to increasing rates of their reactions by the S N 1E1 mechanism. Explain your reasoning. (b) Br -C=CH, A Cl -CH-CH3 A Br | -CH-CH3 B B Br C Cl...
-
What does the following code fragment print? int[] a = { 1, 2, 3 }; int[] b = { 1, 2, 3 }; System.out.println(a == b);
-
Working backward through the statement of cash flows Quinta Company presents the balance sheet shown in Exhibit 5.34 and the statement of cash flows shown in Exhibit 5.35 for 2008. The firm sold...
-
For the system shown below, what should the distance X be for the system to be in equilibrium. 6 lbs 6 lbs 7 lbs 8 lbs 3ft 3ft 2ft Xft
-
The Tesla Roadster, a production electric car, has a \(375 \mathrm{~V}\) battery system that can provide a power of \(200 \mathrm{~kW}\). At this peak power, what is the current supplied by the...
-
Azen Company reported net income of $190,000 for 2012. Azen also reported depreciation expense of $35,000 and a loss of $5,000 on the sale of equipment. The comparative balance sheet shows an...
-
Global Gourmet Coffee Company (GGCC) is a distributor and processor of different blends of coffee. The company buys coffee beans from around the world and roasts, blends, and packages them for...
-
Terfenadine is an antihistaminic drug that contains two alcohol functional groups. Suppose terfenadine were to undergo acid-catalyzed alcohol dehydration (Sec. 10.2). Which alcohol would dehydrate...
-
Explain why two products are formed in the first ether synthesis, but only one in the second. (1) OH i (2) OH + EtOH + NaH HSO4 Etl OEt OEt + OEt
-
Consider the following six tasks that must be assigned to four workers on a conveyor-paced assembly line (i.e., a machine-paced line flow). Each worker must perform at least one task. Time to...
-
Myrtle is fully supported by her three children and Fred, a close friend of the family. Mark paid $4,000 toward Myrtle's support, while Nancy, Opel, and Fred paid $3,200, $800, and $2,000,...
-
Reg. 1.212-1 (n) refers to two Code Sections. What are they?
-
Faraway Travel, Inc. granted its vice-president, Chris Best, an incentive stock option on 1,000 shares of Faraway stock at $25 a share, its fair market value, on July 22, Year 1. Chris exercised the...
-
Determine the amount of self-employment tax due for 2019, assuming that Lynette earned $30,000 in wages and also earned $20,000 from a business that she owned.
-
Julian and Georgia file a joint return. They have adjusted gross income of $112,000 and ritemized deductions of $24,600. Are they Arequired to use the Tax Rate Schedules or the Tax Tables in...
-
Suppose an ANOVA has been performed on a completely randomized design containing six treatment levels. The mean for group 3 is 15.85, and the sample size for group 3 is eight. The mean for group 6 is...
-
When a company has a contract involving multiple performance obligations, how must the company recognize revenue?
-
Explain how you could differentiate between the compounds in each of the following pairs by using simple physical or chemical tests that give readily observable results, such as obvious solubility...
-
Explain why a mixture of two isomeric ethers is formed in the following reaction. NaBH
-
Predict the absolute configuration of the major diol product formed by treatment of (S)-2 ethyl- 2-methyloxirane with water in the presence of an acid catalyst.
-
Carla Vista Hot Dog Stand had the following results last month: Sales revenues were $6200, variable costs were $3100, and fixed costs were $1550 per month. If Carla Vista wants to achieve a targeted...
-
The large computer company you work for just purchased a leading headphone manufacturer. As part of the merger, you have been put in charge of evaluating their manufacturing process. At the time of...
-
A spring gun with spring constant 4 N / m is compressed 2 meters from its natural length and then shoots a ball of 0 . 0 1 kg . What is the speed of the ball out of the gun ( in m / s ) ?

Study smarter with the SolutionInn App


