Terfenadine is an antihistaminic drug that contains two alcohol functional groups. Suppose terfenadine were to undergo acid-catalyzed
Question:
Terfenadine is an antihistaminic drug that contains two alcohol functional groups. Suppose terfenadine were to undergo acid-catalyzed alcohol dehydration (Sec. 10.2).
Which alcohol would dehydrate most rapidly? Why? What would be the dehydration product?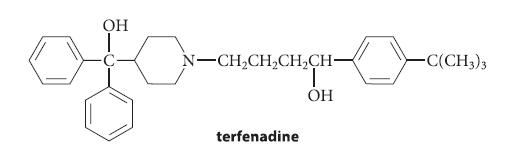
Transcribed Image Text:
OH N-CH₂CH₂CH₂CH- terfenadine Т OH -C(CH3)3
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 100% (1 review)
The question is essentially WhichOH group when protonated and lost as water Sec 102A gives the more ...View the full answer

Answered By

Muhammad Umair
I have done job as Embedded System Engineer for just four months but after it i have decided to open my own lab and to work on projects that i can launch my own product in market. I work on different softwares like Proteus, Mikroc to program Embedded Systems. My basic work is on Embedded Systems. I have skills in Autocad, Proteus, C++, C programming and i love to share these skills to other to enhance my knowledge too.
3.50+
1+ Reviews
10+ Question Solved
Related Book For 

Question Posted:
Students also viewed these Sciences questions
-
List three specific parts of the Case Guide, Objectives and Strategy Section (See below) that you had the most difficulty understanding. Describe your current understanding of these parts. Provide...
-
a firm that wishes to minimize risk when investing idle cashwould be least likely to buy 1. commercial paper 2 long-term corporate bonds c. certifictes of deposit d. treasury bills of the U.S....
-
Managing Scope Changes Case Study Scope changes on a project can occur regardless of how well the project is planned or executed. Scope changes can be the result of something that was omitted during...
-
Write a program HowMany that takes a variable number of command-line arguments and prints how many there are.
-
Interpreting the statement of cash flows Exhibit 5.36 presents a statement of cash flows for Swoosh Shoes Inc., which we derived from actual reports of Nike, Inc., maker of athletic shoes, for three...
-
Corporate engineering of a large corporation usually assumes control of all plant expansion projects in each of its plants for all projects over $25 million. For each case below, discuss the...
-
One design challenge for a capacitor-powered electric car is that the voltage would change with time as the capacitors discharged. If the capacitors in a car were discharged to half their initial...
-
Rainmaker Company prepares its financial statements in accordance with IFRS. In 2012, Rainmaker recorded the following revaluation adjustments related to its buildings and land: The Companys building...
-
On January 1, 2024, for $17.3 million, Marker Company issued 8% bonds, dated January 1, 2024, with a face amount of $19.3 million. For bonds of similar risk and maturity, the market yield is 10%....
-
Predict which of the following compounds should undergo the more rapid reaction with K + (CH 3 ) 3 CO, explain your reasoning, and give the product of the reaction. -CHBr A -CHBr B
-
Arrange the following alcohols according to increasing rates of their acid-catalyzed dehydration to alkene (smallest rate first), and explain your reasoning. ON- CH0- CH0- A B C -CHCHPh T OH CH3 T...
-
Suppose that the last dividend paid by a company was $ 2.20, dividends are expected to grow at a constant rate equal to 5 percent forever, and stockholders require 16 percent to invest in similar...
-
Cal Corey and his wife are living in Las Vegas, Nevada, and own an apartment house from which they receive $20,000 a year in net rental income. Cal's wife also owns an apartment house, which she...
-
Refer to Blacks Law Dictionary or a similar legal reference book and ascertain the definition of the following words or terms: annotated, certiorari, remanded, dictum, acquiesced.
-
Leah Sarah is employed at Cleveland Inc. at an annual salary of $45,000. Leah received $35,000 in salary in 2019 from Cleveland Inc., a prosperous company. They credited the unpaid salary to Leah...
-
An individual performed special valuable services not called for by his employment, in consideration for which his employer agreed to pay him a bonus in an amount to be determined later. After due...
-
Richard and Sally Murphy were divorced in 2017. By the terms of their decree Richard pays Sally $1,000 a month for alimony and child support. When Jane, their 14-year-old daughter, reaches age 18,...
-
A completely randomized design has been analyzed by using a one-way ANOVA. There are four treatment groups in the design, and each sample size is six. MSE is equal to 2.389. Using = .05, compute...
-
Revol Industries manufactures plastic bottles for the food industry. On average, Revol pays $76 per ton for its plastics. Revol's waste-disposal company has increased its waste-disposal charge to $57...
-
Draw the structure of each of the following. (Some parts may have more than one correct answer.) (a) A nine-carbon ether that can be prepared by the Williamson synthesis. (b) An ether that would...
-
Give the products of the reaction of 2-ethyl- 2-methyloxirane (or other compound indicated) with each of the following reagents. (a) Water, H3O+ (b) Water, NaOH, heat (c) NA+ CH3O- in CH3OH (d) CH3OH...
-
Which of the ring-opening reactions given in Fig. Pl 1.48 should occur most readily? Explain.
-
A 39-cm-diameter wheel accelerates uniformly about its center from 110 rpm to 340 rpm in 4.0 s Part A Determine its angular acceleration. Express your answer using two significant figures. Submit...
-
1: Which of the following statements about private banking is true? Private banking focuses on financial transactions that help clients access different markets. Private banking refers to banking...
-
How much of the withdrawal will Murray receive when he makes his minimum annual withdrawal from a RRIF? 100% 100% minus income tax calculated on the basis of his marginal tax rate 100% minus the...

Study smarter with the SolutionInn App


