Draw curved-arrow mechanisms for the reactions given in Fig. P18.78. (a) (b) N=C Figure P18.78 -F +
Question:
Draw curved-arrow mechanisms for the reactions given in Fig. P18.78.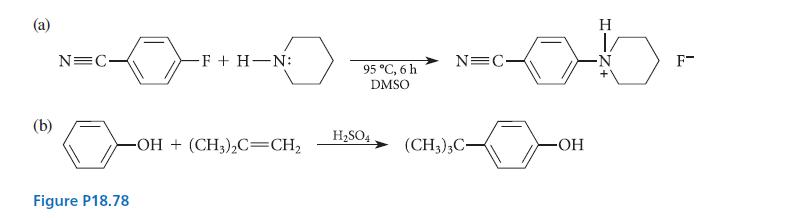
Transcribed Image Text:
(a) (b) N=C Figure P18.78 -F + H-N: -OH(CH3)2C=CH₂ 95 °C, 6 h DMSO H₂SO4 N=C- (CH3)3C- H ob. -OH F-
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 50% (2 reviews)
a b This is a nucleophilic aromatic substitution reaction Notice that the cyano ...View the full answer

Answered By

Sarfraz gull
have strong entrepreneurial and analytical skills which ensure quality tutoring and mentoring in your international business and management disciplines. Over last 3 years, I have expertise in the areas of Financial Planning, Business Management, Accounting, Finance, Corporate Finance, International Business, Human Resource Management, Entrepreneurship, Marketing, E-commerce, Social Media Marketing, and Supply Chain Management.
Over the years, I have been working as a business tutor and mentor for more than 3 years. Apart from tutoring online I have rich experience of working in multinational. I have worked on business management to project management.
5.00+
3+ Reviews
10+ Question Solved
Related Book For 

Question Posted:
Students also viewed these Sciences questions
-
Draw curved-arrow mechanisms for the reactions given in Fig. P18.73.
-
Give curved-arrow mechanisms for the reactions given in Fig. P19.64. (b) (c) S PhCPh (CH3O) 3P: + HC-CH-CH3 CH,CH,NH, + (d) H CH3 HO CH O Figure P19.64 Ph-C-Ph + HS HCI O=CH CH O acid H3C CH3 H CH3...
-
Write out the steps in the reaction mechanisms for the reactions given in eq. 8.20. CHO_ HOCH2CH2OCH, 2-methoxyethanol H+ CH-CH HOCHCH OH HOCH CH2OCH CH OH diethylene glycol
-
Alpha Appliance Service had net income for the year of $ 35,000. In addition, the balance sheet reports the following balances: Calculate the return on assets (ROA) for Alpha Appliance Service for...
-
Refer to the data in Short Exercise 3-10. Requirement 1. Compute Famous Cuts net income for the year ended December 31, 2012.
-
A doctor is getting sued for malpractice by four of her former patients. It is believed that the amount that each patient will sue her for is normally distributed with a mean of $800,000 and a...
-
True or False: If MARR \(=10 \%\) and IRR \(=9 \%\), then ERR \( <10 \%\) and \(\mathrm{PW}>\$ 0\).
-
The ledger of Costello Company at the end of the current year shows Accounts Receivable $110,000, Sales Revenue $840,000, and Sales Returns and Allowances $20,000. Instructions (a) If Costello uses...
-
explain diagram of ER model and relational model and attach their diagrams too Each cinema is identified by its name and has its residency at an address which consists of a street and city only....
-
In some Pd(0)-catalyzed reactions, a Pd(II) compound such as PdCl 2 can be used instead of Pd(0), but it is assumed that the Pd(II) is reduced to Pd(0) in the reaction. Give both the product and the...
-
(a) 1,3-Cyclopentadiene undergoes a DielsAlder reaction with itself to give a diene known commonly as endo-dicyclopentadiene. Give the structure of this diene. (b) When endo-dicyclopentadiene is...
-
In Exercises 6768, graph each semiellipse 2 y = -4-4x
-
How can companies in controversial industries (e.g., tobacco, alcohol, and gambling) achieve a balance between their social responsibility and profit-seeking objectives?
-
Describe what does waiter bring food and beverages to the customer at Air Canada and how each one helps the customers and their actions.?
-
How can casinos promote responsible gambling? Provide some actionable recommendations
-
How can an employer or human resource manager manage mental health issues and illnessin the workplace? Include individual and organizational primary, secondary & tertiary interventions as well as...
-
Explain what these concepts are and how they impact on the different work roles across the community services sector.
-
Garcia, Inc. uses a job-order costing system for its products, which pass from the Machining Department, to the Assembly Department, to finished-goods inventory. The Machining Department is heavily...
-
For each of the following reactions, express the equilibrium constant: a) H20 (I) H2 (g) + 02 (g) Ke = 1.0x107 b) Fe2 (g) 2F (g) Ke= 4.9 x 10-21 c) C (s) + O2 (g) d) H2 (g) + C2H4 (g) C2H6 (g) Ke =...
-
The acid-catalyzed aldol condensation of acetone (just shown) also produces some 2, 6-dimethylhepta-2, 5-dien-4-one. Give a mechanism that explains the formation of this product.
-
Heating acetone with sulfuric acid leads to the formation of mesitylene (1, 3, 5-trimethylbenzene). Propose a mechanism for this reaction.
-
(a) Provide a mechanism for the aldol addition of propanal shown here. (b) How can you account for the fact that the product of the aldol addition is 3-hydroxy- 2-methylpentanal and not...
-
What is specified by the FAA in the Special Condition for the Embraer ERJ 190-300 Airplane entitled "Flight Envelope Protection: High Incidence Protection System" in terms of Capability and...
-
Part 1 Describe the differences between the fiber and matrix materials shown below. For example, discuss the orders of magnitude difference between the material properties, identify how you would...
-
Given: Ref: Gulfstream G450 Altitude = 8000 feet stp MSL S=950 ft; span = 77.9 ft Max Takeoff Gross Wt = 73,900 lbs @ Sea Level: Vstall = 110 KTAS flaps up = 100 KTAS @ 60 deg flaps Best glide speed,...

Study smarter with the SolutionInn App


