Draw the structure of the major species present in solution when 0.01 mole of the following acid
Question:
Draw the structure of the major species present in solution when 0.01 mole of the following acid in aqueous solution is treated with 0.01 mole of NaOH. Explain.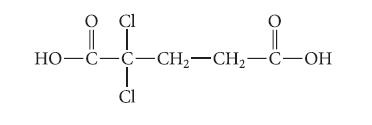
Transcribed Image Text:
O Cl O i OH HO-C-C-CH₂-CH₂-C-OH T CI
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 100% (2 reviews)
Only enough NaOH is present to ionize one of the carboxy ...View the full answer

Answered By

Nazrin Ziad
I am a post graduate in Zoology with specialization in Entomology.I also have a Bachelor degree in Education.I posess more than 10 years of teaching as well as tutoring experience.I have done a project on histopathological analysis on alcohol treated liver of Albino Mice.
I can deal with every field under Biology from basic to advanced level.I can also guide you for your project works related to biological subjects other than tutoring.You can also seek my help for cracking competitive exams with biology as one of the subjects.
3.30+
2+ Reviews
10+ Question Solved
Related Book For 

Question Posted:
Students also viewed these Sciences questions
-
Draw the structure of an optically active triglyceride containing one equivalent of stearic acid and two equivalents of oleic acid. Draw the products expected when this triglyceride reacts with the...
-
When d-glucose is treated with an aqueous bromine solution (buffered to a pH of 6), an aldonic acid is formed called d-gluconic acid. Treatment of d-gluconic acid with an acid catalyst produces a...
-
When the (R, R) isomer of the amine shown is treated with an excess of methyl iodide, then silver oxide, then heated, the major product is the Hofmann product. (a) Draw the structure of the major...
-
Jack Hammer invests in a stock that will pay dividends of $2.00 at the end of the first year; $2.20 at the end of the second year; and $2.40 at the end of the third year. Also, he believes that at...
-
Suppose KC Toys buys $185,800 worth of MegoBlock toys on credit terms of 2/10, n/30. Some of the goods are damaged in shipment, so KC Toys returns $18,530 of the merchandise to MegoBlock. Requirement...
-
Why have FIs been very active in loan securitization issuance of pass-through securities while they have reduced their volume of loan sales? Under what circumstances would you expect loan sales to...
-
What is the expectations gap?
-
On July 31, 2012, Mexico Company paid $3,000,000 to acquire all of the common stock of Conchita Incorporated, which became a division of Mexico. Conchita reported the following balance sheet at the...
-
You are to PAY 350,000 on March 16. Todays spot rate is $1.15/. The forward rate is $1.18/. On March 16 the spot rate is $1.14/. Futures contracts are for 125,000 each. Q1. How many contracts do you...
-
Draw the structures and give the names of all the dicarboxylic acids with the formula C 6 H 10 O 4 . Indicate which are chiral, which would readily form cyclic anhydrides on heating, and which would...
-
Give the product expected when butyric acid (or other compound indicated) reacts with each of the following reagents. (a) Ethanol (solvent), H 2 SO 4 catalyst (b) Aqueous NaOH solution (c) LiAlH 4...
-
In 2010, there were 232.2 million cell phone subscribers in the United States. By 2020, the number of subscribers increased to 276.7 million. a. What is the geometric mean annual percent increase for...
-
The Problems of Multitasking An eastern U.S. financial services company found itself way behind schedule and over budget on an important strategic program. Both the budget and schedule baselines had...
-
Identify changes in two variables that would shift the supply curve of dollars to the right. Identify changes in two variables that would shift the demand curve for dollars to the right.
-
A 0.5-kg cart moving on a horizontal frictionless track at 0.6 m/s hits a solid bumber and bounces back with a speed of 0.4 m/s. The impulse on the cart is?
-
Last year many employers were offering sign up or early bonuses, free luch program, gas cards, flexible work time etc. many perks to their employees. If your employer offered you some of these perks,...
-
Explain what happens to the inflation, unemployment, and output gap in the short run in each of the following circumstances. do not need to graph but explain which part of IS/MP/PC is affected and...
-
A-1 products manufacture wooden furniture using an assembly line process. All direct material are introduced at the start of the process, and conversion cost is incurred evenly throughout...
-
Michelles trust is subject to 3.8% surtax on the lesser of the trusts net investment income or the excess of the trusts adjusted gross income over the $12,400 threshold (the highest trust tax rate)....
-
Furan possesses less aromatic character than benzene as measured by their resonance energies (96 kJ mol-1 for furan; 151 kJ mol-1 for benzene). What reaction have we studied earlier that shows that...
-
If benzene were 1,3,5-cyclohexatriene, the carbon-carbon bonds would be alternately long and short as indicated in the following structures. However, to consider the structures here as resonance...
-
For each of the pairs below, predict specific aspects in their 1H NMR spectra that would allow you to distinguish one compound from the other. (a) (b) (c) Br Br
-
P that (15 pts) Consider the fixed temperature ratio Brayton cycle and the pressure ratio optimizes (maximizes) the work output. Consider now that the compression process has two parts: i. isentropic...
-
1. Define 5D in your own words. If it was defined as an operation that uses "trochoidal passes with a big and tightly controlled maximal radial engagement", what operation is being referred to? 2. Is...
-
Problem 3 Consider the plant described by: i = x + y=[13] a 1. Find the transfer function of the system. 2. Find the closed-loop characteristic equation if the feedback is given by (a) u = - [k k2] x...

Study smarter with the SolutionInn App


