Explain each of the following observations. (a) The optical rotations of alanine are different in water, 1
Question:
Explain each of the following observations.
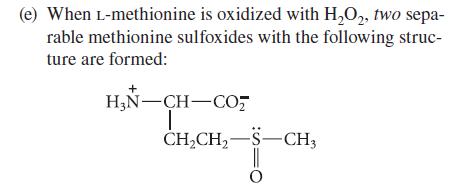
Transcribed Image Text:
(a) The optical rotations of alanine are different in water, 1 M HCl, and 1 M NaOH. (b) Two mono-N-acetyl derivatives of lysine are known. (c) The peptide Gly-Ala-Arg-Ala-Glu is readily hydro- lyzed by trypsin in water at pH = 8, but it is inert to trypsin in 8 M urea at the same pH. (d) After peptides containing cysteine are treated with HSCH₂CH₂OH, then with aziridine, they can be cleaved by trypsin at their (modified) cysteine residues. NH H₂C-CH₂ aziridine
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 100% (1 review)
a b c d Alanine like the other amino acids is a different compound in HCI NaOH and neutral HO because its ionization state is different and different ...View the full answer

Answered By

Muhammad Umair
I have done job as Embedded System Engineer for just four months but after it i have decided to open my own lab and to work on projects that i can launch my own product in market. I work on different softwares like Proteus, Mikroc to program Embedded Systems. My basic work is on Embedded Systems. I have skills in Autocad, Proteus, C++, C programming and i love to share these skills to other to enhance my knowledge too.
3.50+
1+ Reviews
10+ Question Solved
Related Book For 

Question Posted:
Students also viewed these Sciences questions
-
List and explain three (3) reasons why it is important to comply with the Privacy Act (1988) and the Australian Privacy S Principles when gathering information on children and when writing and...
-
This Comprehensive Problem is to acquaint you with the content of the 2009 financial state-ments of Home Depot, Inc. , reproduced in Appendix A of this textbook. (The 2009 financial statements are...
-
This Comprehensive Problem is to acquaint you with the content of the 2012 financial statements of Home Depot, Inc., reproduced in Appendix A of this textbook. (The 2012 financial statements are for...
-
Jane Grimes, retail fruit and vegetable merchant, does not keep a full set of accounting records. However, the following information has been produced from the business's records: 1. Summary of the...
-
What type of depreciation expense pattern is used under each of the following methods and when is its use appropriate? a. The straight-line method. b. The units-of-production method. c. The...
-
You plan to start saving for your sons college education. He will begin college when he turns 18 years old and will need $4,000 then and in each of the following three years. You will make a deposit...
-
Jupiter's is considering an investment in time and administrative expense on an effort that promises one large payoff in the future, followed by additional expenses over a 10-year horizon. The cash...
-
1. Zachary Porter of Highland Heights, Kentucky, is contemplating borrowing $10,000 from his bank. The bank could use add-on rates of 6.5 percent for 3 years, 7 percent for 4 years, and 8 percent for...
-
A wise guy majoring in math says that he thinks your lottery is a bad deal.What other utility might you tell the wise guy he might receive that makes the lottery worth playing?
-
When a mixture of the amino acids Phe and Gly is subjected to chromatography in a pH 6 buffer on the ion-exchange resin shown in Eq. 27.9, the Phe emerges from the column much later than the Gly,...
-
(a) What reagent would be used to convert the corresponding chloromethyl polystyrene resin into the following resin? (b) To a column containing this resin suspended in a pH 6 buffer is added a...
-
Calculate the mass (in g) of each sample. a. 5.94 * 10 20 SO 3 molecules b. 2.8 * 10 22 H 2 O molecules c. 1 glucose molecule (C 6 H 12 O 6 )
-
Sweaters Inc., a sweater manufacturer contracts with Blouse Barn, a clothing store, to sell 20 blue sweaters to Blouse Barn. Sweaters, Inc. delivers 20 purple sweaters. The sweaters were quite...
-
We are all observing consequences of the Supreme Court's holding in Dobbs v. Jackson Women's Health Organization . Explain: (1) how the holding in Dobbs changes the legal standard that applies to...
-
April Showers built a house for Maye Flowers but Maye complained about the finished house. Maye claimed that the garage ceiling was too low and might not accommodate a large SUV; that the patio did...
-
Joshua is a contractor that agrees to build a new commercial office building in downtown Santa Barbara. He begins construction on the building but quickly discovers that he did not purchase the...
-
Company A just paid a dividend of $2.50 per share. Then, it expects its dividend to grow at 6% for the next 3 years. What is the present value of dividends over the next 3 years if the required rate...
-
Seeking to find gainful part-time employment, Sarah Spencer is choosing between two job offers. The first, a sales position at a department store, pays a flat salary of $8 per hour. As Sarah plans to...
-
Prove that if Σ an is absolutely convergent, then a. an
-
Classify each of the labeled bonds in the following structure in terms of the bond type ( or ) and the component orbitals that overlap to form the bond. (For example, the carbon--carbon bond in...
-
Give the structure of: 2E,7Z)-5-[(4- 1-propenyl]-2,7 -non adiene Be sure to read Study Guide Link 4.2 if you have difficulty with this problem.
-
Starting with the same two alkenes, would the products be different if DBr were used? Explain.
-
Assume that the system has a multiple level queue with a variable time quantum per queue, and that the incoming job needs 175 ms to run to completion. If the first queue has a time quantum of 10 ms...
-
What did you think of your practicum? Has it prepared you for the working world?
-
Find all functions g such that g'(x) = f(x). f(x)=2/x6 -2/5x5+ a for x>0 g(x) = 2/5x5+b for x <0 g(x) = -2x-5/5+c O 2/5x5+a for x>0 g(x)= 2/5x5 + b for x < 0 -2/5x5+a for x>0 g(x) = -2/5x5+b for x <0

Study smarter with the SolutionInn App


