Explain why each alkyl halide stereoisomer gives a different alkene in the E2 reactions shown in Fig.
Question:
Explain why each alkyl halide stereoisomer gives a different alkene in the E2 reactions shown in Fig. P9.80. It will probably help to build models or draw out the conformations of the two starting materials.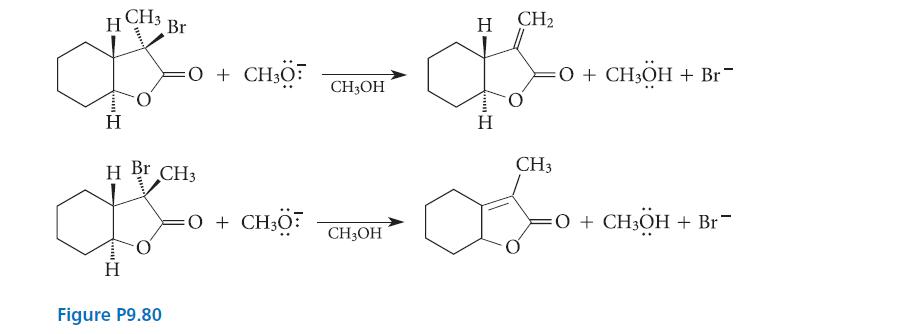
Transcribed Image Text:
H¹ Br **** H H Br CH3 H =O + CH3O Figure P9.80 =O + CH3O CH3OH CH3OH H CH₂ H =O + CH3ÖH + Br CH3 af =O + CH3ÖH + Br
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 100% (3 reviews)
a The key to the solution is to assume that E2 reactions are faster ...View the full answer

Answered By

Mugdha Sisodiya
My self Mugdha Sisodiya from Chhattisgarh India. I have completed my Bachelors degree in 2015 and My Master in Commerce degree in 2016. I am having expertise in Management, Cost and Finance Accounts. Further I have completed my Chartered Accountant and working as a Professional.
Since 2012 I am providing home tutions.
3.30+
2+ Reviews
10+ Question Solved
Related Book For 

Question Posted:
Students also viewed these Sciences questions
-
Planning is one of the most important management functions in any business. A front office managers first step in planning should involve determine the departments goals. Planning also includes...
-
Choose the alkyl halide(s) from the following list of C 6 H 13 Br isomers that meet each criterion below. (1) 1-bromohexane (2) 3-bromo-3-methylpentane (3) 1-bromo-2,2-dimethylbutane (4)...
-
1) As a result of the US Senate elections in Georgia on January 5, 2021, Democrats now hold majority in both chambers of the US congress, which will likely lead to a significant increase in...
-
Food Enterprises is analyzing the performance of their retail business and have calculated Value At Risk at $17,350,000 under the statistical method at a 95% confidence level, and a mean of $898,000....
-
On July 1, 2011, the City of Belvedere accepted a gift of cash in the amount of $3,000,000 from a number of individuals and foundations and signed an agreement to establish a private-purpose trust....
-
Sentinel Systems Inc. prepared the following sales budget for the current year: At the end of December 2014, the following unit sales data were reported for the year: For the year ending December 31,...
-
True or False: If \(\operatorname{IRR}(\mathrm{A})>\operatorname{IRR}(\mathrm{B})\), then \(\operatorname{ERR}(\mathrm{A})>\operatorname{ERR}(\mathrm{B})\).
-
The following data, presented in alphabetical order, are taken from the records of Radar Corporation. Accounts payable................... $ 240,000 Accounts receivable.................. 140,000...
-
On June 30, 2016, Flint Limited issued 13.75% bonds with a par value of $802,000 due in 20 years. They were issued at 99 and were callable at 102 at any date after June 30, 2023. Because of lower...
-
The cis and trans stereoisomers of 4-chlorocyclohexanol give different products when they react with OH , as shown in the reactions given in Fig. P9.83. (a) Give a curved-arrow mechanism for the...
-
(a) Tell whether each of the eliminations shown in Fig. P9.79 is syn or anti. (b) Reaction (2) shows first-order kinetics. Draw a curved-arrow mechanism for this reaction that is consistent with its...
-
(a)Prove that an orthogonal 2 ( 2 matrix must have the form Where Is a unit vector. (b)Using part (a), show that every orthogonal 2 ( 2 matrix is of the form Where 0 ( ( ( 2 (. or cos sin -sin cos...
-
Examine the principles of organization in the light of criticisms.
-
Distinguish between devious and legitimate political tactics in organizations.
-
What is meant by corporate or organizational culture and how does it differ from organizational climate?
-
Comment on an open system of organization, with reference to socio-technical systems.
-
What are the organizational factors that contribute to political behaviour?
-
J. Morgan of SparkPlug Inc. has been approached to take over a production facility from B.R. Machine Company. The acquisition will cost $1,500,000, and the after-tax net cash inflow will be $275,000...
-
A fuel pump sends gasoline from a car's fuel tank to the engine at a rate of 5.88 10-2 kg/s. The density of the gasoline is 735 kg/m3, and the radius of the fuel line is 3.18 10-3 m. What is the...
-
Identify the chirality centers in the following molecules (yellow-green = Cl, pale yellow =F): (b) (a) Threose (a sugar) Enflurane (an anesthetic)
-
Is cocaine (Worked Example 9.2) dextrorotatory or levorotatory?
-
A 1.50 g sample of coniine, the toxic extract of poison hemlock, was dissolved in 10.0mL of ethanol and placed in a sample cell with a 5.00 cm path length. The observed rotation at the sodium D line...
-
16. If the following statement were used in a C++ program, what would it cause to be written on the screen? cout < < "C++ is easy to understand.";
-
Function main() of a C++ program is: int main ( ) { int marks[100], numStudents, average, best, m; cout < >numStudents; Obtain( marks, numStudents); cout <
-
Sigma Corporation applies overhead cost to jobs on the basis of direct labor cost. Job V , which was started and completed during the current period, shows charges of $ 6 , 8 0 0 for direct...

Study smarter with the SolutionInn App


