Explain why the following compound does not undergo base-catalyzed exchange in D 2 O even though it
Question:
Explain why the following compound does not undergo base-catalyzed exchange in D2O even though it has an a-hydrogen.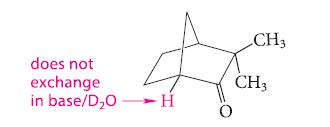
Transcribed Image Text:
does not exchange in base/D₂OH O CH3 CH3
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 100% (QA)
Exchange does not occur because the orbital containing the unshared electron pair on the acarbo...View the full answer

Answered By

John Kago
Am a processional practicing accountant with 5 years experience in practice, I also happens to have hands on experience in economic analysis and statistical research for 3 years. am well conversant with Accounting packages, sage, pastel, quick books, hansa world, etc, I have real work experience with Strata, and SPSS
4.70+
31+ Reviews
77+ Question Solved
Related Book For 

Question Posted:
Students also viewed these Sciences questions
-
The following compound is not aromatic even though it has 4n + 2 electrons in a continuous cyclic array. Explain why this compound is not aromatic.
-
Explain why the following alkyl halide does not undergo a substitution reaction, regardless of the conditions under which the reaction is run: Cl
-
The bicycle ketone shown below does not undergo aldol self-condensation even though it has two ? hydrogen atoms. Explain.
-
Suppose that General Motors Acceptance Corporation issued a bond with 10 years until maturity, a face value of $1000, and a coupon rate of 7% (annual payments). The yield to maturity on this bond...
-
G Wholesale Company began the year with inventory of $6,000. During the year, G purchased $97,000 of goods and returned $6,200 due to damage. G also paid freight charges of $1,500 on inventory...
-
The financial statements for BSW National Bank (BSWNB) are shown below: a. What is the dollar value of earning assets held by BSWNB? b. What is the dollar value of interest-bearing liabilities held...
-
Xie Company identified the following activities, costs, and activity drivers for 2017. The company manufactures two types of go-karts: deluxe and basic. Required 1. Compute a single plantwide...
-
The stockholders equity accounts of Karp Company at January 1, 2014, are as follows. Preferred Stock, 6%, $50 par ................$600,000 Common Stock , $5 par .................... 800,000 Paid-in...
-
A system consists of a large number of identical molecules at equilibrium. Each molecule can be in one of a ladder of energy levels. As shown in the diagram below, the energy levels are uniformly...
-
Sketch the proton NMR spectrum of 2-butanone, and explain how this spectrum would change if the compound were treated with D 2 O and a base.
-
Outline a malonic ester synthesis of the following carboxylic acid: CH3 COH 2-methylheptanoic acid
-
The file EuroTourism2 contains a sample of 28 European countries. Variables included are the number of jobs generated in the travel and tourism industry in 2015, the spending on business travel...
-
Where is the image located when an object is placed 25 cm in front of a convex mirror with a focal length of 11 cm?
-
We supply 132 J of heat to an engine with 11.2 % efficiency. (a) How much work (J) can the engine do? (b) How much heat (J) must the engine exhaust?
-
Compute the total work done by a gas system shown in figure below following expansion processes: P (KPA) 500- 0 A 0.3 B 0.6 PV.4 = C 0.9 V (m)
-
1. Consider the following algorithm: Algorithm rec Fun(x, y, n) ifn <1 else if n-1 return Null return x + y else return (x + y) * rec Fun(x, y, n-1) a. What is the base condition? b. How do the...
-
A nearsighted man uses his ideally prescribed eyeglasses that have a refractive power of -2.20 diopters. He would like a prescription for contact lenses. What is the correct contact lens prescription...
-
Refer to the original data given for Jupiter Game Company in the preceding problem. An activity-based costing study has revealed that Jupiters $150,000 of fixed costs include the following...
-
Portal Manufacturing has total fixed costs of $520,000. A unit of product sells for $15 and variable costs per unit are $11. a). Prepare a contribution margin income statement showing predicted net...
-
Provide the reagents necessary to accomplish the following syntheses. (a) (b) (c) (d) MeO MeO SEt SEt
-
Provide reagents that would accomplish the follwing syntheses. (a) (b) HO Glycerol Epichlorohydrin
-
Write structures for compounds A-J showing stereochemistry where appropriate. (a) What is the stereochemical relationship between A and C? (b) (c) What is the stereochemical relationship between H...
-
Evaluate the integrals: (a) Z -dz = (b) / 2 12 dz -j to z2 = j that except for the end where C is any contour from 21 points, lies on the left side of the y-axis.
-
A restaurant received an 18% discount on a purchase of fifteen 1.14 L. Bottles of Wine, selling for 12.50 per bottle. What is the total cost with the discount plus HST (13%)
-
If the estimated standard error of the mean for low income respondents is 0.26, the margin of error at a confidence level of 95% is _. Report the number rounded to the fourth decimal place (0.1234)...

Study smarter with the SolutionInn App


