Outline a malonic ester synthesis of the following carboxylic acid: CH3 COH 2-methylheptanoic acid
Question:
Outline a malonic ester synthesis of the following carboxylic acid: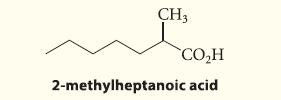
Transcribed Image Text:
CH3 CO₂H 2-methylheptanoic acid
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 50% (2 reviews)
Using the analysis in the text identify the acetic acid unit in the carboxylic acid T...View the full answer

Answered By

Gilbert Chesire
I am a diligent writer who understands the writing conventions used in the industry and with the expertise to produce high quality papers at all times. I love to write plagiarism free work with which the grammar flows perfectly. I write both academics and articles with a lot of enthusiasm. I am always determined to put the interests of my customers before mine so as to build a cohesive environment where we can benefit from each other. I value all my clients and I pay them back by delivering the quality of work they yearn to get.
4.80+
14+ Reviews
49+ Question Solved
Related Book For 

Question Posted:
Students also viewed these Sciences questions
-
What alkyl bromide(s) should be used in the malonic ester synthesis of each of the following carboxylic acids? a. Propanoic acid b. 2-methylpropanoic acid c. 3-phenylpentanoic acid d....
-
a. What carboxylic acid would be formed if the malonic ester synthesis were carried out with one equivalent of malonic ester, one equivalent of 1,5-dibromopentane, and two equivalents of base? b....
-
Outline all steps in a malonic ester synthesis of each of the following: (a) pentanoic acid, (b) 2-methylpentanoic acid, (c) 4-methylpentanoic acid.
-
Suppose Targets stock has an expected return of 20% and a volatility of 40%, Hersheys stock has an expected return of 12% and a volatility of 30%, and these two stocks are uncorrelated. a. What is...
-
Shine King Cleaning has decided that, in addition to providing cleaning services, it will sell cleaning products. During December, Shine King completed the following transactions: Dec 2 Purchased 600...
-
Megalopolis Bank has the following balance sheet and income statement. For Megalopolis, calculate: a. Return on equity b. Return on assets c. Asset utilization d. Equity multiplier e. Profit margin...
-
Refer to the information in Exercise 16-14. Prepare journal entries dated June 30 to record: (a) raw materials purchases, (b) direct materials usage, (c) indirect materials usage, (d) direct labor...
-
Plant-wide, department, and activity-cost rates Allen's Aero Toys makes two models of toy airplanes, fighter jets, and cargo planes. The fighter jets are more detailed and require smaller batch...
-
Consider two oppositely charged atoms with charges of +1 and -1 units, respectively. The two atoms interact with each other through electrostatic (Coulombic) and van der Waals forces, and reach an...
-
Explain why the following compound does not undergo base-catalyzed exchange in D 2 O even though it has an a-hydrogen. does not exchange in base/DOH O CH3 CH3
-
Write a mechanism involving an enolate ion intermediate for the reaction shown in Eq. 22.6. Explain why only the a-hydrogens are replaced by deuterium. H H H L DO/dioxane Et3N: (a base) heat, 48 h D...
-
Gaseous NO 2 decomposes at 573 K. The concentration of NO 2 was measured as a function of time. A graph of 1/[NO 2 ] versus time gives a straight line with a slope of 1.1 L/mol s. What is the rate...
-
what are the environmental factors if tim hortons open in ecuador?
-
Based on the spot speed data collected 50.0 km/h 55.4 km/h 60.0 km/h 64.4 km/h O 70.0 km/h OO If the density at 48% occupancy is 60 veh/km for a specific lane of a road, what would be the density at...
-
What is the change in internal energy of a system that takes in 3.83 10^6 J of heat, and does 4.50 10^5 J of work while dissipating 9.67 10^6 J of heat?
-
Omega Inc. is a large business with several divisions, including Delta Division.Omegas headquarters and the majority of its operations are located in Canada, and are concentrated in natural resource...
-
What role does value chain analysis play in competitive advantage?
-
Jupiter Game Company manufactures pocket electronic games. Last year Jupiter sold 25,000 games at $25 each. Total costs amounted to $525,000, of which $150,000 were considered fixed. In an attempt to...
-
The Higher the time period of the financial security the higher the. ............... risk. O a. Maturity O b. Default and Maturity Oc. Default O d. Liquidity
-
Write a mechanism that accounts for the following reaction: OH HA + HOH
-
Propose a reasonable mechanism for the following reaction. cat. H2SO4 OH- C3
-
Propose a reasonable mechanism for the following reaction. OH
-
6. (15 pts.) Here's a dataset I just generated. I'm not going to tell you what distribution I generated the data from because......I don't want to. 1.491, 0.019, 0.318, 0.056, 0.816, 0.978, 0.174,...
-
If the estimated standard error of the mean for low income respondents is 0.26, what is the upper limit of a confidence interval for a confidence level of 95%? Round to the second decimal place...
-
8. Use the Matrix method to solve algebraically the given a) (4 pts.){ x-y -2x + 3y = 10 -21

Study smarter with the SolutionInn App


