From what alkene could each of the following glycols be prepared by the OsO 4 or KMnO
Question:
From what alkene could each of the following glycols be prepared by the OsO4 or KMnO4 method?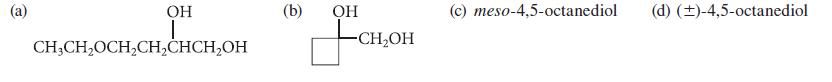
Transcribed Image Text:
OH I CH3CH₂OCH₂CH₂CHCH₂OH (b) OH -CH₂OH (c) meso-4,5-octanediol (d) (±)-4,5-octanediol
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 100% (QA)
a b The alkene required is 4ethoxy1butene CH3CHOCH...View the full answer

Answered By

OTIENO OBADO
I have a vast experience in teaching, mentoring and tutoring. I handle student concerns diligently and my academic background is undeniably aesthetic
4.30+
3+ Reviews
10+ Question Solved
Related Book For 

Question Posted:
Students also viewed these Sciences questions
-
From which alkene could each of the following cyclopropane derivatives be prepared using the Simmons- Smith reaction? CH3
-
Planning is one of the most important management functions in any business. A front office managers first step in planning should involve determine the departments goals. Planning also includes...
-
What alkene should be used to synthesize each of the following alkyl bromides? a. b. c. d. CHa CH3CCH Br CH2CHCH3 Br CH3 CCH 2 CH2CH3
-
1. Calculate the budgeted nights booked: Maximum capacity (30 rooms) * Number of days per year (365) * Expected occupancy rate (80%) = 8760 nights. 2. Calculate the tariff revenues: Budgeted nights...
-
Do all transactions by U.S. companies with foreign parties require special accounting procedures by the U.S. companies? Explain.
-
Describe three systems development tools and three development methods.
-
Consider the Gallo strategic decision. Describe how you would go about evaluating that decision.
-
TufStuff, Inc., sells a wide range of drums, bins, boxes, and other containers that are used in the chemical industry. One of the companys products is a heavy-duty corrosion-resistant metal drum,...
-
An aluminum-alloy rod has a length of 9.2293 cm at 20.00C and a length of 9.2767 cm at the boiling point of water. (a) What is the length of the rod at the freezing point of water? (b) What is the...
-
Show a curved-arrow mechanism for the first step, and the structure of the cyclic intermediate formed, when an alkene is treated with KMnO 4 . A Lewis structure for the permanganate ion is as...
-
Predict the major product(s) of each of the following transformations. (a) Et C-CH + CHOH (solvent) ***** H Et (optically active) HSO4 (trace) HSO4 (b) The enantiomer of the epoxide in part (a) +...
-
You must have completed Exercise 11 of Chapter 10 before beginning this exercise, and thus have used the SQL Data Definition Language to populate the tables for the three relations COMPANY, STUDENT,...
-
Using the information given in question 10, compute SSR, SSE, SST, and R 2 . Also use an F-test to test the significance of the model.
-
Use the foregoing information to construct 95 % confidence intervals for the parameters.
-
For the return data with NYSE Index being explained by Ford and Chrysler, suppose we expect the returns for Ford and Chrysler are 0.0229 and 0.0337, respectively. Forecast the return for NYSE and...
-
When heteroscedasticity is detected, we sometimes use a weighted regression in which the dependent and independent variables are weighted by the variances of their error terms. Thus, the estimated...
-
A cylinder of a radioactive solid, 1.5 cm in radius and 2 m long, is encased in a cylindrical shell of pyrex glass, 3.0 cm in outside radius. The radioactive core generates heat at the steady rate of...
-
Explain how the capital recovery concept applies to the taxation of annuities. Consider both purchased annuities and pension payments in your answer.
-
Reconsider Prob. 1474. In order to drain the tank faster, a pump is installed near the tank exit as in Fig. P1475. Determine how much pump power input is necessary to establish an average water...
-
Cardiolipins arc a group of lipids found in heart muscles. What products would be formed if all ester bonds, including phosphates, were saponified by treatment with aqueousNaOH? ROCH2 CH2OOR" A...
-
Stearolic acid, C18H32O2 yields stearic acid on catalytic hydrogenation and undergoes oxidative cleavage with ozone to yield nonanoic acid and nonanedioic acid. What is the structure of Stearolic...
-
How would you synthesize Stearolic acid (Problem 27.19) from 1-dccyne and 1-chloro-7-iodohcptane?
-
Suppose you visit a small branch of Bank of Sultanate in South Mawaleh that operates a single server with an exponential service time that has rate 1 transaction every 5 minutes. Arrivals at this...
-
In July 2020, a brawl involving approximately 12 adults took place in the hotel lobby of the Seminole Hard Rock Hotel & Casino in Florida.6 Although unproved at the time of writing, it is alleged...
-
Provide a statement explaining your own personal code of ethics. Include at least 4 codes in your answer with enough explanation and examples. In addition, discuss whether the personal code of ethics...

Study smarter with the SolutionInn App


