Predict the major product(s) of each of the following transformations. (a) Et C-CH + CHOH (solvent) *****
Question:
Predict the major product(s) of each of the following transformations.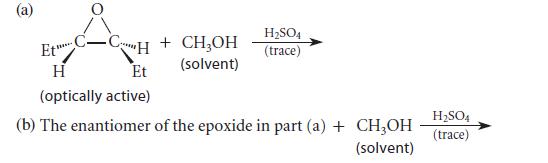
Transcribed Image Text:
(a) Et C-CH + CH₂OH (solvent) ***** H Et (optically active) H₂SO4 (trace) H₂SO4 (b) The enantiomer of the epoxide in part (a) + CH3OH (trace) (solvent)
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 100% (3 reviews)
a b The nucleophilic reaction of methanol on the protonated epoxide occurs with inversion to give 3...View the full answer

Answered By

PALASH JHANWAR
I am a Chartered Accountant with AIR 45 in CA - IPCC. I am a Merit Holder ( B.Com ). The following is my educational details.
PLEASE ACCESS MY RESUME FROM THE FOLLOWING LINK: https://drive.google.com/file/d/1hYR1uch-ff6MRC_cDB07K6VqY9kQ3SFL/view?usp=sharing
3.80+
3+ Reviews
10+ Question Solved
Related Book For 

Question Posted:
Students also viewed these Sciences questions
-
Predict the major product from each of the following reactions. (a) (b) (c) (d) (e) (f) (g) (h) (i) CH NH2, cat. HA N-H, cat. HA NH2 cat. HA PPha (1) HS SH (2) Raney Ni, H2 0 CH2PPha (excess) O. O...
-
Predict the major product from each of the following reactions. (a) (b) (c) (d) (e) (f) OH SOC, pyr OH HBr NaNH2 OH OH (1) TsCI, pyr (2) EtSNa Nal, H2SO OH
-
Predict the major product from each of the following reactions. (a) (b) (c) (d) (e) HO OH NH2 HA (cat.) (1) HCN (2) LiAIH4 (3) H2o mCPBA
-
Question 13 Case Study - Little Pear Administration Pty Ltd (LPA) You are the payroll officer for Little Pear Administration Pty Ltd (LPA). Debra Foy is a company employee and has approached you with...
-
Why dont the additions and deductions from the bank balance on a bank reconciliation require adjustment by the company?
-
Should the IT director report to the company president, or somewhere else? Does it matter?
-
In question 1, identify the marketing as opposed to the business strategy. Data From Question 1: What is a business strategy? Do you agree with the definition proposed? Illustrate your answer with...
-
Tiger Computers, Inc., of Singapore is considering the purchase of an automated etching machine for use in the production of its circuit boards. The machine would cost $900,000. (All currency amounts...
-
Use the attached image and SQL DDL Commands to query the appropriate tables and populate with four rows of data. Please include explanation for script. CUSTOMER PK CustomerID CustomerName Address...
-
From what alkene could each of the following glycols be prepared by the OsO 4 or KMnO 4 method? OH I CH3CHOCHCHCHCHOH (b) OH -CHOH (c) meso-4,5-octanediol (d) ()-4,5-octanediol
-
Explain each of the following facts with a mechanistic argument. (a) When butyl methyl ether (1-methoxybutane) is treated with HI and heat, the initially formed products are mainly methyl iodide and...
-
Radio waves, microwaves, and infrared all belong to the electromagnetic radiation spectrum. These terms reference ranges of radiation frequencies we use every day in our wireless networking...
-
The thermal conductivity of a certain solid material is k = 25 W/m K. At a certain point and time in a block of this material sketched in Fig. P.1.1, the temperature is 65C, the temperature gradient...
-
One of the main sources of water pollution is agricultural runoff. Consider three options to control this problem: (1) a CAC requirement prohibiting irrigation ditches from draining directly into...
-
Suppose that you are in charge of the demand-side management program at Megabucks Power, and your state public utility commission has just decided that you can now count energy-efficiency measures as...
-
Governor Blabla has decided that, rather than build a new nuclear power plant to service power needs, the state should save an equivalent amount of energy. As one component of an efficiency plan, he...
-
Reducing the rate of infant and childhood mortality in poor countries clearly leads to a short-run increase in population growth. Use the economic model of family size developed in this chapter to...
-
How is the gross income from a rental property or a royalty property determined?
-
Beginning with a country that has a trade deficit, demonstrate graphically what will happen to a countrys potential output with globalization if that countrys costs of production fall. Explain your...
-
Show the products you would expect to obtain from reaction of glyceryl trioleate with the following reagents: (a) Excess Br2 in CH2Cl2, (b) H2/Pd (c) NaOH/H2C (d) O3, then Zn/CH3CO2H (e) LiAlH4, then...
-
How would you convert oleic acid into the following substances? (a) Methyl oleate (b) Methyl stearate (c) Nonanal (d) Nonanedioic acid (e) 9-Octadccynoic acid (Stearolic acid) (f) 2-Bromostearic acid...
-
Cold-water fish like salmon are rich in omega-.3 fatty acids, which have a double bond three carbons in from the non carboxyl end of the chain and have been shown to lower blood cholesterol levels....
-
Regardless of whether or not you plan to be an HR professional, how might you use this competency model for your professional development? Specifically, how would you go about gaining proficiency in...
-
i. Briefly explain what a stakeholder is? ii. Give 2 examples of stakeholders with regards to a health care system. iii. Explain why each example from part ( ii ) are stakeholders?
-
For a Fixed Price Incentive Fee (FPIF) contract with the following details: Negotiated Target Cost = $225,000 Target Fee (Profit) = $40,000 Ceiling Price = $300,000 Cost Sharing Ratio (CSR) = 60/40...

Study smarter with the SolutionInn App


