Give the product(s) expected when each of the following compounds is treated with periodic acid. OH OH
Question:
Give the product(s) expected when each of the following compounds is treated with periodic acid.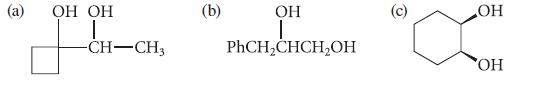
Transcribed Image Text:
З OH OH | -CH-CH3 (b) ОН | PhCH₂CHCH₂OH ОН OH
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 100% (2 reviews)
a O CHCH3 T b c C...View the full answer

Answered By

Vincent Omondi
I am an extremely self-motivated person who firmly believes in his abilities. With high sensitivity to task and operating parameters, deadlines and keen on instructions, I deliver the best quality work for my clients. I handle tasks ranging from assignments to projects.
4.90+
109+ Reviews
314+ Question Solved
Related Book For 

Question Posted:
Students also viewed these Sciences questions
-
Give the product expected when each of the following compounds is treated with acid. CO.H CO,H + heat)
-
Predict the major product obtained when each of the following compounds is treated with bromine (Br 2 ) together with sodium hydroxide (NaOH) followed by aqueous acid (H 3 O + ). (a) (b) (c)
-
Draw the enolate that is formed when each of the following compounds is treated with LDA. (a) (b) (c) (d)
-
Identify any one non governmental / non-profit organization in Toronto locality. Brief introduction to the organization that includes the following information: Vision/Mission Services / Programs...
-
Refer to PE 6-64. Make all journal entries necessary on the companys books to adjust the reported cash balance in response to the receipt of the bank statement. Data from PE 6-64 Bank Reconciliation...
-
The income statement for the year ended December 31, 2011, and the balance sheets at December 31, 2010, and December 31, 2011, for Craigs Service Company are presented here. Requirements 1. Prepare a...
-
What performance measure would you consider most important for McDonald's? For Chevrolet?
-
(Settlement of Debt) Strickland Company owes $200,000 plus $18,000 of accrued interest to Moran State Bank. The debt is a 10-year, 10% note. During 2010, Stricklands business deteriorated due to a...
-
You have just been named Director of Data Administration of General Hardware Co. General Hardware maintains a large, central IS organization with several operational relational databases at its...
-
What glycol undergoes oxidation to give each of the following sets of products? (a) HC HC C=0+0= (b) O
-
Show a curved-arrow mechanism for the first step, and the structure of the cyclic intermediate formed, when an alkene is treated with KMnO 4 . A Lewis structure for the permanganate ion is as...
-
Gruneisen constant (a) Show that the free energy of a phonon mode of frequency w is k B T in [2sinh (hw/2k B T)]. It is necessary to retain the zero-point energy hw to obtain this result. (b) If is...
-
Extend the analysis conducted in Problem 11.8 by relaxing the assumption that the solute concentration C A, s remains constant. In a practical applications, C A, s will vary as a result of adsorption...
-
Water at 25C flows at 5 ft/s inside a straight cylindrical tube made of benzoic acid, with a 1-in. inside diameter. If the tube is 20 ft long, estimate the mixing-cup average concentration of benzoic...
-
At the same time that wild salmon are threatened with extinction across the planet, salmon is now widely available and relatively cheap in U.S. supermarkets. The reason? The explosive growth of...
-
Estimate the thermal conductivity of liquid isopropyl alcohol at 300 K and compare your estimates with the experimental value available in Section 2 of Perrys Chemical Engineers Handbook.
-
One economic development paradox receiving recent research attention has been the dismal economic performance of many mineral-dependent countries in the Third World. On average, they have fared very...
-
What effect does an asset's adjusted basis have in determining the gain or loss realized upon its sale?
-
Using (1) or (2), find L(f) if f(t) if equals: t cos 4t
-
Propose a biosynthetic pathway for the sesquiterpene helminthogermacrene from farnesvldiphosphate.
-
Identify the following fatty acid, and tell whether it is more likely to be found in peanut oil or in redmeat:
-
Fats can be either optically active or optically inactive, depending on their structure. Draw the structure of an optically active fat that yields 2 equivalents of stearic acid and 1 equivalent of...
-
If a project with a duration of 1 year had an actual cost of $30,000, a planned cost of $50,000, and a value completed of $25,000 at the end of 6 months, what are the CPI, SPI and CSI for the project?
-
Analyse how McDonald's has navigated the dynamic nature of culture in its global marketing strategies. Specifically, discuss how the company's adaptation to local cultural nuances influences consumer...
-
Differentiate between McDonald's targeting strategy for children, young adults, and health-conscious consumers. How has the corporation's differentiated targeting strategy influenced its product...

Study smarter with the SolutionInn App


