Give the structure of an aldehyde C 8 H 8 O 2 that would be oxidized to
Question:
Give the structure of an aldehyde C8H8O2 that would be oxidized to terephthalic acid by KMnO4.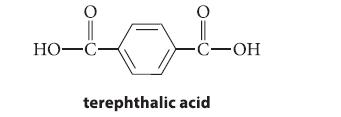
Transcribed Image Text:
HO-C- O -C-OH terephthalic acid
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 100% (QA)
The starting material has five degrees of ...View the full answer

Answered By

Ankit Mahajan
I am an electrical engineering graduate from Thapar institute of engineering and technology.
Qualified exams - GATE 2019,2020.
CAT EXAM 2021- 91.4 percentile
SSC EXAMS- 2019,2020,2021
AFCAT EXAM- 2019,2020,2021
I want to share my knowledge with other people so that they can achieve the same.
I have strong hold Mathematics, Electrical engineering and all the subjects related.
Just give me a problem and I will give you the solution of it.
5.00+
1+ Reviews
10+ Question Solved
Related Book For 

Question Posted:
Students also viewed these Sciences questions
-
Give the structure of an intramoleczlar substitution product and an intermolecular substitution product that might be obtained from 4-bromo-l-butanol on treatment with one equivalent of NaOH. Which...
-
Give the structure of the product derived by intramolecular aldol condensation of the keto aldehyde shown: O CHI KOH. H-0 CH CCH CCHOCHIoO CH3
-
Give the structure of the product derived by intramolecular aldol condensation of the keto aldehyde shown:
-
According to a study by the American Pet Food Dealers Association, 63% of U.S. house-holds own pets. A report is being prepared for an editorial in the San Francisco Chronicle. As a part of the...
-
XM, Ltd., was a small engineering firm that built hi-tech robotic devices for electronics manufacturers. One very complex device was partially completed at the end of 2014. Barb McLauren, head...
-
Consider the following discrete probability distribution. a. Is this a valid probability distribution? Explain. b. Graphically depict this probability distribution. c. What is the probability that...
-
When the Concepcions went to AT&T to get a phone, they were confused as to why they were paying money for the phone when it was advertised as being free. They had to pay sales tax on the full normal...
-
Quigley Corporations trial balance at December 31, 2014, is presented below. All 2014 transactions have been recorded except for the items described below. Unrecorded transactions 1. On January 1,...
-
The kibitzer computes the average of these three probabilities and announces the result of his computation publicly. Beatrice and Carol update their probabilities for F in the light of this new...
-
Give the products expected (if any) when acetone reacts with each of the following reagents. (a) HO+ (b) NaBH4 in CHOH, then HO (c) CrO3, pyridine (d) NaCN, pH 10, HO (e) CH3OH (excess), HSO4 (trace)...
-
Outline a Wittig synthesis for each of the following alkenes; give two Wittig syntheses of the compound in part (a). CH0- -CH=CH- (mixture of cis and trans) (b) CH3 I HC=CCHCH3 CHCH=
-
Facepalm, Instarant, and Snaphat are rival firms in an oligopoly industry. If kinked-demand theory applies to these three firms, Facepalms demand curve will be: a. more elastic above the current...
-
Estimate the total cost and cost per square foot for the installation of face bricks and CMU's required in the building shown in the attached diagram. Wall ties are placed 16 in. c.c. in the...
-
Visit the web pages: Transparency Org Write n essay-type summary: Investigate the different levels of corruption in countries and explain the form used to determine the corruption rate. Mention the...
-
Standard Bank Group To Reveal 2025 Ambition During Virtual Strategy Update For Investors And Analysts Standard Bank Group ("Standard Bank") will host a strategic update event via webcast for...
-
What are your most difficult tasks in preparing your financial plan? Why?
-
Establishing a Shared Service Centre in China A global consulting company decided to centralize some of its finance and human resource-related activities from its Asia-Pacific operations in China. An...
-
Kitchen Kings Toledo plant manufactures three product lines, all multi-burner, ceramic cook tops. The plants three product models are the Regular (REG). the Advanced (ADV), and the Gourmet (GMT)....
-
Is the modified 5-question approach to ethical decision making superior to the modified moral standards or modified Past in approach?
-
Diacyl peroxides decompose readily when heated. (a) What factor accounts for this instability? (b) The decomposition of a diacyl peroxide produces CO2. How is it formed? (c) Diacyl peroxides are...
-
Write a structural formula for each of the following compounds: (a) Octanoic acid (b) Propanamide (c) N, N-Diethylhexanamide (d) 2-Methyl-4-hexenoic acid (e) Butanedioic acid (f) 1, 2-Benzenedioic...
-
Give an IUPAC systematic or common name for each of the following compounds: (a) (b) (c) (d) (e) (f) (g) (h) CH3CN Cl NH2
-
Your competitor serves a complete shrimp dinner for $19.95. You would like to meet this offer, but you're not sure you can afford to. You know that your overhead is 38% of sales, labor is 30% of...
-
Problem 4. (a) Determine the equivalent stiffness for the figure below as a single spring system. k. F k k3 Figure. Multiple springs in series and parallel (b) Analyze forces/moments on the systems...
-
The pinion in a gear mesh has 25 teeth and the mating gear has 124. Both are cut to a module of 2 mm, a face width of 100 mm, and an AGMA quality number of A10. The drive is a commercial enclosed...

Study smarter with the SolutionInn App


