How many chemically nonequivalent sets of hydrogens are in each of the following structures? (a) CH3CH (c)
Question:
How many chemically nonequivalent sets of hydrogens are in each of the following structures?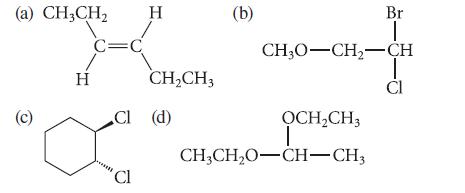
Transcribed Image Text:
(a) CH3CH₂ (c) H C=C CI Cl Cl H CH₂CH3 (d) (b) CH3O-CH₂-CH ỌCH₂CH3 Br T CH3CH₂O-CH-CH3 CI
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 100% (1 review)
a c CH3CH three chemically nonequivalent sets of ...View the full answer

Answered By

Grace Igiamoh-Livingwater
I am a qualified statistics lecturer and researcher with an excellent interpersonal writing and communication skills. I have seven years tutoring and lecturing experience in statistics. I am an expert in the use of computer software tools and statistical packages like Microsoft Office Word, Advanced Excel, SQL, Power Point, SPSS, STATA and Epi-Info.
5.00+
1+ Reviews
10+ Question Solved
Related Book For 

Question Posted:
Students also viewed these Sciences questions
-
(a) Show that replacing each of the CH2 protons by some group Q in the (S) enantio-mer of 2-butanol leads to a pair of diastereomers, as it does for the (R) enantiomer. (b) How many chemically...
-
Using the equations your derived in questions 59 and 62 for IS and LM, what will be the equilibrium interest rate (r) and equilibrium output (Y)? A) B) C) D) r = 8, Y = 900B r = 9, Y = 600B r = 10, Y...
-
How many hydrogens are replaced by deuterium when each of the following compounds is treated with NaOD in D2O? a. 3-methylcyclopentanone b. 3-methylhexanal
-
Using the table of implied volatilities below ("volatility surface"), calculate the implied volatility a trader would use for pricing an 8-month option with K/S0 = 1.04. Hint: Interpolate linearly. 1...
-
In the year 2002, Salt Lake City, Utah, hosted the Winter Olympics. To get ready for the Olympics, most of the major roads and highways in and around Salt Lake City were renovated. It took over three...
-
Lev, in his article On the Usefulness of Earnings (1989), pointed out the low ability of reported net income to explain variations in security prices around the release date of earnings information....
-
Using the variance data for Menounos Manufacturing Co., prepare an income statement through gross profit for the year ended December 31, 20Y6. Assume the company sold 3,000 units at $100 per unit.
-
The Marlin Company produces plastic bottles to customer order. The quality inspector randomly selects four bottles from the bottle machine and measures the outside diameter of the bottle neck, a...
-
Research Web-based database technologies and identify a database management system (other than SQL Server, MySQL, or Oracle) that is used to deploy applications to the Web and the cloud. Discuss the...
-
(a) How many electrons are involved in the oxidation of triphenylphosphine (Ph 3 P;) to triphenylphosphine oxide (Ph 3 P = O)? Show your reasoning. (b) Draw a resonance structure for...
-
How many electrons are involved in the oxidation of 1-propanethiol to each of the following compounds. (See Fig. 10.3 for detailed Lewis structures.) (a) 1-propanesulfonic acid, CH 3 CH 2 CH 2 SO 3 H...
-
Determine the pressure drop across a 34-in diameter Type L copper pipe that is 60 ft long and is carrying water at 12 gpm.
-
Trexon plc is a major oil and gas exploration business that has most of its operations in the Middle East and South-East Asia. Recently, the business acquired rights to explore for oil and gas in the...
-
Describe how foreign currency options can be used for hedging in the situation so that (a) ImportCo is guaranteed that its exchange rate will be less than 1.4600, and (b) ExportCo is guaranteed that...
-
A stock index currently stands at 350 . The risk-free interest rate is \(8 \%\) per annum (with continuous compounding) and the dividend yield on the index is \(4 \%\) per annum. What should the...
-
Refer to Exercise 12-22. 1. Calculate the amount and cost of (a) unused manufacturing capacity and (b) unused selling and customer-service capacity at the beginning of 2013 based on actual production...
-
When you open a can of soda (or beer), compressed CO2 expands irreversibly against the atmosphere as it bubbles up through the drink. Assume that the process is adiabatic and that the CO2 has an...
-
Sam is an executive with a U.S. corporation. During the current year, he is working in another country. His employer provides a corporation-owned residence for Sam that is located three miles from...
-
Find the velocity, acceleration, and speed of a particle with the given position function. r(t) = (t 2 , sin t - t cos t, cos t + t sin t), t > 0
-
Identify the following intermediate in the citric acid cycle, and tell whether it has R or Sstereochemistry:
-
The following compound is an intermediate in the biosynthesis of one of the twenty common a-amino acids. Which one is it likely to be, and what kind of chemical change must take place to complete...
-
The following compound is an intermediate in the pentose phosphate pathway, an alternative route for glucose metabolism. Identify the sugar it is derivedfrom.
-
Capital Budgeting Case / Problem Parameters: A capital project and budgeting decision is being considered that would involve an expansion, along with a replacement of some old equipment. The project...
-
Difference in standard deviation and beta as measures of risk Security Expected Return Standard Deviation G 14.0% 20% H 12.0% 28% Which security has the least total risk? Which has the least...
-
Each of two independent projects has a probability 0.98 of a loss of $1 million and 0.02 probability of a loss of $10 million What is the 97.5% VaR for each project? What is the 97.5% expected...

Study smarter with the SolutionInn App


