Identify all of the asymmetric carbon atoms (if any) in each of the following structures. (a) (b)
Question:
Identify all of the asymmetric carbon atoms (if any) in each of the following structures.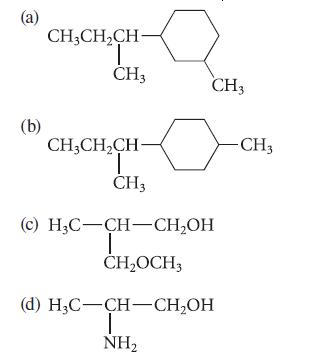
Transcribed Image Text:
(a) (b) CH₂CH₂CH- T CH3 CH3CH₂CH- T CH3 CH3 (c) H₂C-CH-CH₂OH T CH₂OCH3 (d) H₂C-CH-CH₂OH T NH₂ -CH3
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 100% (3 reviews)
The asymmetric carbons are indicated ...View the full answer

Answered By

Ashington Waweru
I am a lecturer, research writer and also a qualified financial analyst and accountant. I am qualified and articulate in many disciplines including English, Accounting, Finance, Quantitative spreadsheet analysis, Economics, and Statistics. I am an expert with sixteen years of experience in online industry-related work. I have a master's in business administration and a bachelor’s degree in education, accounting, and economics options.
I am a writer and proofreading expert with sixteen years of experience in online writing, proofreading, and text editing. I have vast knowledge and experience in writing techniques and styles such as APA, ASA, MLA, Chicago, Turabian, IEEE, and many others.
I am also an online blogger and research writer with sixteen years of writing and proofreading articles and reports. I have written many scripts and articles for blogs, and I also specialize in search engine
I have sixteen years of experience in Excel data entry, Excel data analysis, R-studio quantitative analysis, SPSS quantitative analysis, research writing, and proofreading articles and reports. I will deliver the highest quality online and offline Excel, R, SPSS, and other spreadsheet solutions within your operational deadlines. I have also compiled many original Excel quantitative and text spreadsheets which solve client’s problems in my research writing career.
I have extensive enterprise resource planning accounting, financial modeling, financial reporting, and company analysis: customer relationship management, enterprise resource planning, financial accounting projects, and corporate finance.
I am articulate in psychology, engineering, nursing, counseling, project management, accounting, finance, quantitative spreadsheet analysis, statistical and economic analysis, among many other industry fields and academic disciplines. I work to solve problems and provide accurate and credible solutions and research reports in all industries in the global economy.
I have taught and conducted masters and Ph.D. thesis research for specialists in Quantitative finance, Financial Accounting, Actuarial science, Macroeconomics, Microeconomics, Risk Management, Managerial Economics, Engineering Economics, Financial economics, Taxation and many other disciplines including water engineering, psychology, e-commerce, mechanical engineering, leadership and many others.
I have developed many courses on online websites like Teachable and Thinkific. I also developed an accounting reporting automation software project for Utafiti sacco located at ILRI Uthiru Kenya when I was working there in year 2001.
I am a mature, self-motivated worker who delivers high-quality, on-time reports which solve client’s problems accurately.
I have written many academic and professional industry research papers and tutored many clients from college to university undergraduate, master's and Ph.D. students, and corporate professionals. I anticipate your hiring me.
I know I will deliver the highest quality work you will find anywhere to award me your project work. Please note that I am looking for a long-term work relationship with you. I look forward to you delivering the best service to you.
3.00+
2+ Reviews
10+ Question Solved
Related Book For 

Question Posted:
Students also viewed these Sciences questions
-
Identify all of the asymmetric carbon atoms in each of the following structures. (a) (b) (c) , , H C CH CH2OH NH2 CH3
-
The following four structures are naturally occurring optically active compounds. Star the asymmetric carbon atoms in these structures. CHO H CH, COOH OH OH H,N H serine erythrose menthol camphor
-
Indicate the asymmetric carbon atoms in the following compounds: (a) (b) CH3 CH3 CH2-CH-CH-C NH2 NH2 Br Br
-
The best measure of a firm's sustainable income is a .income before extraordinary items. b .net income. c. income before extraordinary item and change in accounting principle. d. income from...
-
Did the company cause the harm?
-
Case A. Food Fare is a small chain of restaurants that has developed a loyal customer base by providing fast-food items with more choices (e.g., how the hamburger should be cooked; self-serve...
-
Increasingly, we are seeing email used in cases involving defendants located in foreign countries. Plaintiffs filed suit against four Defendants: Qingdao Sunflare New Energy Co., Skone Lighting Co.,...
-
Operating profits and losses for the seven industry segments of Foley Corporation are: Penley $ 90 Cheng $ (20) Konami (40) Takuhi 34 KSC 25 Molina 150 Red Moon 50 Based only on the operating profit...
-
thank you I've been having lots of troubles opening any documents in full they just appear as blank would you know why this is or perhaps be able to assist me
-
As described in the previous account, Pasteur discovered two stereoisomers of tartaric acid. Draw their structures [you cannot tell which is (1) and which is (2)]. Which stereoisomer of tartaric acid...
-
Arsenic (As) is below nitrogen and phosphorus in Group 5A of the periodic table. In an arsine (R 3 As;) the RAsR bond angles are about 92. How would you expect the inversion rate of arsines to...
-
How could the incentives facing the people at the FDA be changed to reduce the incidence of Type II errors?
-
Portal Doors sells steel entry doors for $285 each. The firm has fixed costs of $380,000 and variable costs per unit of $125. What is Portal's break-even point in sales dollars?
-
Suppose that Lisa purchases 1 pizza per month when the price is $20 and 8 pizzas per month when the price is $13. What is the price elasticity of Lisa's demand curve?
-
Allan is seriously considering terminating his $500,000 whole life policy. The $3,000 annual premium was due 10 days ago but has not yet been paid. The policy's cash surrender value (CSV) is $75,000...
-
Country M is one of the largest fresh milk producers in the world. A recent virus outbreak has caused many cows to get sick, which greatly affected the supply chain for milk. This has resulted in...
-
Pooja's sister (Kiara) became quite ill earlier this year and had extensive medical bills which she could not cover. Kiara's financial problems were made worse as she had to take a 10-month leave of...
-
A mathematically fair bet is one in which a gambler bets, say, $100 for a 10 percent chance to win $1000 ($100 = .10 x $1000). Assuming diminishing marginal utility of dollars, explain why this is...
-
The Home Depot is the leading retailer in the home improvement industry and one of the 10largest retailers in the United States. The company included the following on its January 29, 2012, balance...
-
P-Bromotoluene reacts with potassium amide to give a mixture of m- and p-methyl aniline. Explain.
-
Propose a mechanism to account for the reaction of benzene with 2, 2, 5, 5- tetra methyl tetra hydrofuran. H2SO4
-
Propose a mechanism to account for the following reaction: C CH2CI AICI3
-
A taxpayer is expensing $1,500,000 in equipment and wants to maximize the amount of expensing under Section 179 on her return. What is the total amount of Section 179 expensing that she can claim on...
-
A taxpayer is making some changes to their home by installing some energy-efficient property in TY2023. Last year, they claimed $300 in nonbusiness energy property credit for similar changes. This...
-
A taxpayer is placing a vehicle into service at a cost basis of $45,000 and which is used 100% for business purposes. This vehicle is subject to limits on passenger vehicles under Section 280F. What...

Study smarter with the SolutionInn App


