Propose a mechanism to account for the following reaction: C CH2CI AICI3
Question:
Propose a mechanism to account for the following reaction:
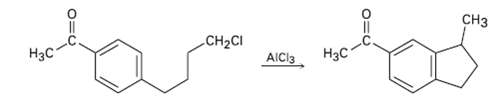
Transcribed Image Text:
Cнз CH2CI Нас Нзс AICI3
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 54% (11 reviews)
H CHCl love formation of H3C0 H3C HC CIAICI3 CH3 HA loss of H AICI 3 CH3 ...View the full answer

Answered By

John Kago
Am a processional practicing accountant with 5 years experience in practice, I also happens to have hands on experience in economic analysis and statistical research for 3 years. am well conversant with Accounting packages, sage, pastel, quick books, hansa world, etc, I have real work experience with Strata, and SPSS
4.70+
31+ Reviews
77+ Question Solved
Related Book For 

Question Posted:
Students also viewed these Organic Chemistry questions
-
Propose a mechanism to account for the reaction of benzene with 2, 2, 5, 5- tetra methyl tetra hydrofuran. H2SO4
-
Propose a mechanism to account for the formation of 3, 5-dimethylpyrazole from hydrazine and 2, 4-pcntancdionc. Look carefully to see what has happened to each carbonyl carbon in going from starting...
-
Propose a mechanism to account for the fact that heating 1-deuterioindene scrambles the isotope label to all three positions on the five-memberedring. 1-Deuterioindene
-
What is the result of the following? A. [Natural History, Science] B. [Natural History, Science, Art] C. The code does not compile. D. The code compiles but throws an exception at runtime. } import...
-
Morgan (age 45) is single and provides more than 50% of the support of Rosalyn (a family friend), Flo (a niece, age 18), and Jerold (a nephew, age 18). Both Rosalyn and Flo live with Morgan, but...
-
Consider a random mixture containing 4.00 g of Na2CO3 (density 2.532 g/mL) and 96.00 gof K2CO3 (density 2.428 g/mL) with a uniform spherical particle radius of 0.075 mm. (a) Calculate the mass of a...
-
1. Which normal curve has a greater mean? 2. Which normal curve has a greater standard deviation?
-
GenX Furnishings Company manufactures designer furniture. GenX Furnishings uses a job order cost system. Balances on June 1 from the materials ledger are as follows:...
-
An employer has scheduled candidates for interviews. When one person is told of her interview time, she says she is unavailable due to caregiving responsibilities, and asks for another time. The...
-
A hydrogen atom (Z = 1) is in the third excited state, and a photon is either emitted or absorbed. Concepts: (i) What is the quantum number of the third excited state? (ii) When an atom emits a...
-
P-Bromotoluene reacts with potassium amide to give a mixture of m- and p-methyl aniline. Explain.
-
In the Gatterman-Koch reaction, a formyl group (?CHO) is introduced directly onto a benzene ring. For example, reaction of toluene with CO and HCl in the presence of mixed CuCl/AlCl 3 gives...
-
Determine whether the 20-lb block shown is in equilibrium, and find the magnitude and direction of the friction force when P = 12.5 lb and = 15. 4= 0.30 A = 0.25 20
-
Compare Alphabet Inc. and Amazon in Revenue Growth from 2018 to 2021? explain with references.
-
Evaluate (8) (9) 5x+6ydydx =
-
23. Given the following HTML mark-up and CSS rules, what is the text colour of the word Rainy? HTML mark-up: Today is a Rainy Day CSS rules: body {color: black; } section, p { color: green; } p...
-
4 (a): An investment plan in perpetual bond is offering an expected return of Rs. 7500 at the end of every 3 months period. If Sheraz requires a nominal rate of return of 12% per year compounded...
-
Lions Furniture received an invoice in the amount of $6,951 dated September 16, 2021 with terms 5/10, 2/20, n/60. If the invoice is to be paid in full on September 27, how much will the payment be?
-
Based on the email that Mukilteo received, the IC members perspective is correct with regard to: A. layering and netting of fees. B. tactical allocation capabilities. C. manager-specific operational...
-
Anna, a high school counselor, devised a program that integrates classroom learning with vocational training to help adolescents at risk for school dropouts stay in school and transition to work...
-
Electrical power is to be produced from a steam turbine connected to a nuclear reactor. Steam is obtained from the reactor at 540 K and 36 bar, the turbine exit pressure is 1.0 bar, and the turbine...
-
Show the resonance structures for the conjugate base of phenol.
-
The pKa for phenol is 10, the pKa for ethanol is 16, and the pKa for carbonic acid (H2CO3) is 6.35. Complete these equations and predict whether the reactants or the products are favored...
-
Provide names for thesecompounds: a) CH;CH,CH,CH,CH,CH b) CH f) CH,CCH,CCH, d) CI g) h)
-
What theorists are most likely to argue the city's form and growth come from decisions made by people and organizations that control wealth and other key resources?
-
Explain the difference between glucogenic and ketogenic amino acids
-
Describe what is an arrangement where workers who don't join a union must make payments equal to union dues and fees to get union representation services?

Study smarter with the SolutionInn App


