In each of the following pairs, one of the glycols is virtually inert to periodate oxidation. Which
Question:
In each of the following pairs, one of the glycols is virtually inert to periodate oxidation. Which glycol is inert? Explain why.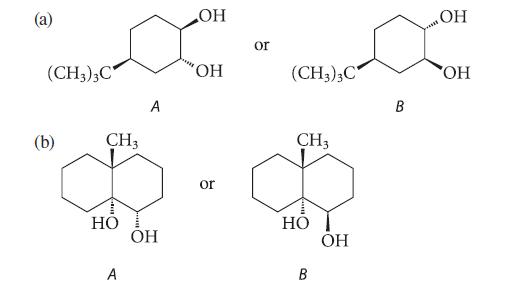
Transcribed Image Text:
В (с (CH3)3C (b) CH3 HO A A OH OH OH or or (CH3)3C CH3 HO B OH B OH OH
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 100% (1 review)
a Because the large tertbutyl group virtually locks the molecule into the chair confo...View the full answer

Answered By

Deborah Joseph
My experience has a tutor has helped me with learning and relearning. You learn everyday actually and there are changes that are made to the curriculum every time so being a tutor has helped in keeping me updated about the present curriculum and all.
I have also been able to help over 100 students achieve better grades particularly in the categories of Math and Biology both in their internal examinations and external examinations.
5.00+
2+ Reviews
10+ Question Solved
Related Book For 

Question Posted:
Students also viewed these Sciences questions
-
In each of the following pairs , one. of the glycols is virtually inert to periodate oxidation. Which glycol is inefi? Explain why. CH3 CH, or
-
Which compound in each of the following pairs would have the higher boiling point? Explain your answers. (a) (b) (c) (d) (e) (f) (g) (h) Hexane, CH3(CH2)4CH3, or nonane, CH3(CH2)7CH3 (i) OH or HO OH...
-
In each of the following pairs of substances, one is stable and known, whereas the other is unstable. For each pair, choose the stable substance, and explain why the other compound is unstable. a....
-
The following question is designed to highlight key concepts from the Loyalty Programs topic article titled, "StarBUCKS, Loyalty, and Breakage" (Nevraumont 2019). Q. Author's position. "If you hire...
-
In an inflationary period, which inventory cost flow method will produce the highest net income? Explain.
-
Multiple-Choice Questions 1. Evans Company completes a service engagement and bills a customer $50,000 on June 19, 2010. Included in the journal entry to record this transaction will be a: a. debit...
-
Monroe Bradstad borrowed \($100,000\) from his aunt, Jeanne Garland, to purchase farmland. Both parties subsequently signed a promissory note stipulating that interest would be accrued prior to or on...
-
You are doing the first-year audit of Sherman School District and have been assigned responsibility for doing a four-column proof of cash for the month of October 2011. You obtain the following...
-
Explain the types of anomalies that might occur on this relational model when not normaliz ed NORMALIZATION (40 Points) Consider a COLLEGE logical relational model below....
-
Draw a curved-arrow mechanism for each of the conversions shown in Fig. P11.77. Figure P11.77 S
-
(a) Give a curved-arrow mechanism for the reaction shown in Fig. P11.70. Be sure your mechanism indicates the role of the weak acid ammonium chloride. (b) Why does the reaction of an aziridine...
-
Under what conditions of temperature and pressures would you expect a real gas to have (a) Z < 1; (b) Z > 1?
-
Identify three separate jobs. These can be current jobs, jobs you have held in the past, and/or jobs that you are familiar with. Use JDS to assess each jobs motivating potential. Formulate a plan to...
-
Assume that the membrane rejection of the protein is over 99% and calculate the membrane rejection of the non-protein constituents in the previous problem.
-
What is the resolution of two Gaussian peaks of identical width (3.27 s) and height eluting at 67.3 s and 74. 9 s, respectively?
-
A certain compound, A, reacts to form products according to the reaction A P. The amount of A is measured as a function of time under a variety of different conditions, and the tabulated results are...
-
Use the data given in exercise 11.35. Follow the instructions given in exercise 11.43. Exercise 11.35. A random sample of people were asked whether they were athletic, moderately athletic (Mod), or...
-
Polly has the following capital gains and losses for the current year: Short-term capital gain$ 1,000 Short-term capital loss 8,000 Long-term capital gain 5,000 Collectibles gain 16,000 Collectibles...
-
An investor sells a European call on a share for $4. The stock price is $47 and the strike price is $50. Under what circumstances does the investor make a profit? Under what circumstances will the...
-
Choline, a component of the phospholipids in cell membranes, can be prepared by SN2 reaction of trimethyl amine with ethylene oxide. Show the structure of choline, and propose a mechanism for...
-
Cyclopentamine is an amphetamine-like central nervous system stimulant. Propose a synthesis of Cyclopentamine from materials of five carbons orless. CH3 -CH2CHNHCH3 Cyclopentamine
-
Tetracaine is a substance used medicinally as a spinal anesthetic during lumbar punctures (spinal taps). (a) How would you prepare tetracaine from the corresponding aniline derivative, ArNH2? (b) How...
-
An insurance broker, whether individual, corporate or partnership, must engage solely in the business of a general insurance broker, but this does NOT apply: a) To members who have been specifically...
-
A paralegal, has been told that if he bills $1,500 hours by January 1 of next year, he will receive a bonus. On December 1, Brad realizes he is within 200 hours of reaching his goal. What are some of...
-
In 2010, the PCAOB's scope of responsibility was expanded to include overseeing audits of securities brokers and dealers by the - Act. (Enter only one word per blank.)

Study smarter with the SolutionInn App


