In each of the following sets, arrange the three compounds in order of increasing reactivity toward base-promoted
Question:
In each of the following sets, arrange the three compounds in order of increasing reactivity toward base-promoted hydrolysis, least reactive first, and explain your reasoning.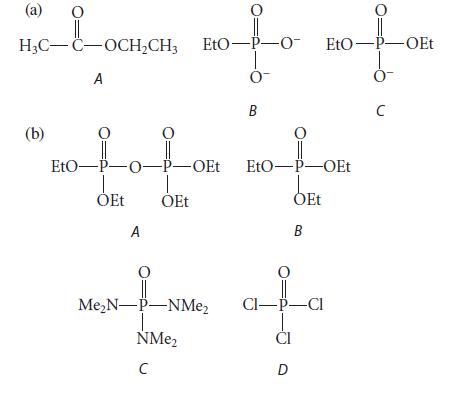
Transcribed Image Text:
(a) nofo H₂C-C-OCH₂CH3 EtO-P-O- A 0- B (b) EtO-P-O-P-OEt ỎEt A OEt O Me₂N-P-NMe₂ NMe₂ C O EtO-P-OEt 0- с EtO-P-OEt OEt B CI-P-CI CI D
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 80% (5 reviews)
a b The order ...View the full answer

Answered By

S Mwaura
A quality-driven writer with special technical skills and vast experience in various disciplines. A plagiarism-free paper and impeccable quality content are what I deliver. Timely delivery and originality are guaranteed. Kindly allow me to do any work for you and I guarantee you an A-worthy paper.
4.80+
27+ Reviews
73+ Question Solved
Related Book For 

Question Posted:
Students also viewed these Sciences questions
-
Arrange the compounds in each of the following sets in order of decreasing pKa, highest first. Explain your reasoning. (a) CLCH2CH2SH CH3CH2OH CH3CH2SH (b) CH,CH,OH (CH3),N-CH-CH,OH (CH3)N OH
-
Arrange the bonds in each of the following sets in order of increasing polarity: (a) C-F, O-F, Be-F (b) O-Cl, S-Br, C-P (c) C-S, B-F, N-O
-
Arrange the compounds in each of the following groups in order of increasing solubility in water, and briefly explain your answers: a. 1-octanol; ethanol; ethyl chloride b. HOCH2(CHOH)3CH2OH;...
-
Suzanne acquired the following ordinary shares in Quarine plc: She made no further acquisitions and the shares were valued at 3.20 each on 31 March 1982. On 24 July 2020, Suzanne sold 1,200 shares...
-
The counting rate from a radioactive source is 6400 counts/s. The half-life of the source is 10 s. Make a plot of the counting rate as a function of time for times up to 1 min. What is the decay...
-
Parker Investments has EBIT of $20,000, interest expense of $3,000, and preferred dividends of $4,000. If it pays taxes at a rate of 38%, what is Parkers degree of financial leverage (DFL) at a base...
-
Discuss the occupational stereotypes that you hold about computer programmers, the clergy, truck drivers, bartenders, and bankers. How do you think these stereotypes have developed? Has an...
-
The comparative balance sheets for Rothlisberger Company as of December 31 are presented below. Additional information: 1. Operating expenses include depreciation expense of $42,000 and charges from...
-
With reference to the following diagram Server/host X Router Y Segment 1 Segment 2 What behavior could this network potentially exhibit? What kind of traffic could cause this behavior?
-
Complete the reactions shown in Fig. P25.24 by drawing the structures of the products, and explain your reasoning. (b) (c) afa CI H3C- + HN(CH,), (large excess) Holofo Figure P25.24 (1) TsCl OH...
-
The anomeric proton (red) in the a-anomer of glucose-1-phosphate in the proton NMR spectrum appears at 5.45. It is split into a doublet of doubletsfour lines of equal sizewith coupling constants of...
-
In Exercises 2532, find the zeros for each polynomial function and give the multiplicity for each zero. State whether the graph crosses the x-axis, or touches the x-axis and turns around, at each...
-
Describe some cognitive or behavioral limitations you have experienced when making consumption decisions and how these limitations can lead to hand-to-mouth consumption. Why is it easier to forecast...
-
Describe some of the historical similarities and differences between recessions. For example, do they always have the same duration and severity? What about expansions?
-
All U.S. states require children to attend school. Mandatory school attendance ends at ages 1618, depending on the state. The adult literacy rate in North America was 100% in 2020. Discuss how...
-
You currently have $10,000 in total wealth and rate your current utility at 4.25. You are deciding if you should invest your money in your friends automotive restoration business. There is a 50%...
-
Over long periods of time, the percentage of households income spent on consumption does not change much even as income rises. Use the five insights into the relationship between income and...
-
A company has 1,250 employees, and you want to take a simple random sample of n = 60 employees. Explain how you would go about selecting this sample by using the table of random numbers. Are there...
-
In Exercises delete part of the domain so that the function that remains is one-to-one. Find the inverse function of the remaining function and give the domain of the inverse function. f(x) = 16x4 -3...
-
When the conjugate-base enolate of diethyl malonate is treated with bromobenzene, no diethyl phenylmalonate is formed. Explain why bromobenzene is inert. CH (CO,Et)2 + Br" diethyl phenylmalonate
-
The reactions of ester enolate ions are not restricted to simple alkylations. With this in mind, suggest the structure of the product formed when the enolate ion formed by the reaction of fm-butyl...
-
Outline a synthesis of each of the following compounds from a -keto ester; then show how the -keto ester itself can be prepared. CHj
-
A disk between vertebrae in the spine is subjected to a shearing force of 580 N. Find its shear deformation taking it to have the shear modulus of 1.00 x10 9 N/m2. The disk is equivalent to a solid...
-
Solve the following initial value problem y" - 24' -8y = t - zu (t-4)+5(t-5) 2u y(0)=0 y6)=0
-
What are the challenges and opportunities associated with the implementation of performance management systems in dynamic and rapidly changing environments, and how can adaptive performance...

Study smarter with the SolutionInn App


