The anomeric proton (red) in the a-anomer of glucose-1-phosphate in the proton NMR spectrum appears at
Question:
The anomeric proton (red) in the a-anomer of glucose-1-phosphate in the proton NMR spectrum appears at δ 5.45. It is split into a doublet of doublets—four lines of equal size—with coupling constants of 3.5 Hz and 7.3 Hz. Explain the origin of each splitting.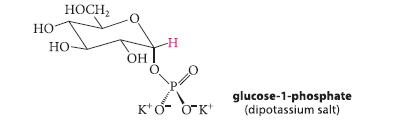
Transcribed Image Text:
HO HOCH₂ HO- OH -H Κ'0 ΟΚ' glucose-1-phosphate (dipotassium salt)
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 100% (1 review)
The anomeric proton is split by both the proton on the adjacent carbon and the phos...View the full answer

Answered By

Sultan Ghulam Dastgir
The following are details of my Areas of Effectiveness English Language Proficiency, Organization Behavior , consumer Behavior and Marketing, Communication, Applied Statistics, Research Methods , Cognitive & Affective Processes, Cognitive & Affective Processes, Data Analysis in Research, Human Resources Management ,Research Project,
Social Psychology, Personality Psychology, Introduction to Applied Areas of Psychology,
Behavioral Neurosdence , Historical and Contemporary Issues in Psychology, Measurement in Psychology, experimental Psychology,
5.00+
1+ Reviews
10+ Question Solved
Related Book For 

Question Posted:
Students also viewed these Sciences questions
-
The 1H NMR data for the two anomers included very comparable peaks in the d 2.0-5.6 region but differed in that, as their highest peaks, anomer V had a doublet at d 5.8 (1H, J = 12 Hz) while anomer...
-
Compound B has the composition 62.1% carbon, 10.3% hydrogen and 27.6% oxygen. Its mass and 1H NMR spectra are shown below. a. b. a. Calculate the empirical formula of B. b. From the mass spectrum,...
-
Managing Scope Changes Case Study Scope changes on a project can occur regardless of how well the project is planned or executed. Scope changes can be the result of something that was omitted during...
-
William made the following acquisitions of preference shares in Pangol plc: In January 2020, the company made a 1 for 4 bonus issue. In February 2021, William sold 450 shares for 4 per share....
-
(a) Show that ke 2 = 1.44 MeV fm, where k is the Coulomb constant and e is the electron charge. (b) Show that hc = 1240 MeV fm.
-
Grey Products has fixed operating costs of $380,000, variable operating costs of $16 per unit, and a selling price of $63.50 per unit. a. Calculate the operating breakeven point in units. b....
-
Using implicit personality theory, explain how physical attractiveness influences job-related outcomes in employment interviews and performance appraisals.
-
Evaluate Walmarts new marketing campaign and tagline. Did the company make the right decision to drop Always Low Prices. Always. as a tagline? Why or why not?
-
How did XYZ Electronics allocate its marketing budget to ensure the successful execution of its strategy?
-
In each of the following sets, arrange the three compounds in order of increasing reactivity toward base-promoted hydrolysis, least reactive first, and explain your reasoning. (a) nofo HC-C-OCHCH3...
-
What would the G be for ATP hydrolysis in a cell in which the concentrations of ATP, ADP, and phosphate were all 1 mM?
-
A group of ten people were selected and given a standard IQ test. These scores were then compared with their high school grades. Find \(r\) and determine whether it is statistically significant at...
-
For each of the following scenarios, identify whether it is best explained as an example of overconfidence, availability bias, anchoring bias, representativeness bias, focusing illusion, or loss...
-
For each of the following transactions, determine if it takes place in the market for outputs or the market for inputs. Then, determine the direction of the flow of goods and services and the flow of...
-
How is macroeconomic equilibrium related to equilibrium output? How does equilibrium output differ from potential output?
-
Sarah, Alicia, and Philip all lost their jobs when the medical billing firm they worked for was acquired by another company. After a few weeks of searching for another full-time job, Sarah decided to...
-
Which of the following people are consumption smoothing and which are not? Explain your reasoning using the opportunity cost and marginal principles. a. Sharon Tirabassi won $10 million in the...
-
A particular automobile costs an average of $17,755 in the Pacific Northwest. The standard deviation of prices is $650. Suppose a random sample of 30 dealerships in Washington and Oregon is taken,...
-
Why did management adopt the new plan even though it provides a smaller expected number of exposures than the original plan recommended by the original linear programming model?
-
Give the product expected when methyl methacrylate (methyl 2-methylpropenoate) reacts with each of the following reagents. (a) C2H5SH and NaOMe catalyst in MeOH (b) NaOH
-
Give a curved-arrow mechanism for each of the following reactions. H2C NaOEt catalyst O Ph SH EtOH CH PhSH2C O (mixture of stereoisomers; why?) CH3
-
Provide structures for the missing nucleophiles that could be used in the following transformations. NaOEt Ho+ CH, CH3
-
Morton Inc's February purchases on account were $100,000. February cash payments to vendors were $80,000. the Accounts Payable balance on February 29, 2020 was $30,000. What was the balance of...
-
Everest Corp. bought a one-year insurance on August 1, 2023 which took effect on the same day. The total cost of the insurance was $7,200 and was paid with cash in full. The total cost was debited to...
-
Toby's Bakery Store uses the accrual basis of accounting. A customer ordered cupcakes on June 5. Toby delivered the cupcakes to the customer on June 7. The customer mailed payment to Toby on June 10....

Study smarter with the SolutionInn App


