Indicate whether the asymmetric atom in each of the following compounds has the R or S configuration.
Question:
Indicate whether the asymmetric atom in each of the following compounds has the R or S configuration.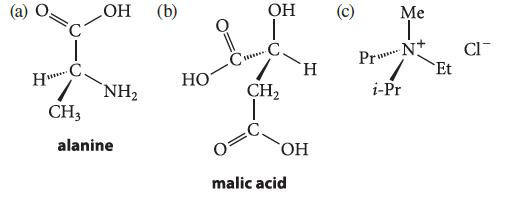
Transcribed Image Text:
H с. www. CH3 OH (b) NH₂ alanine НО www. ОН | CH₂ H OH malic acid О Me I ***** Pr N+ i-Pr Et CIT
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 100% (QA)
a The asymmetric carbon in the given stereoisom...View the full answer

Answered By

Asim farooq
I have done MS finance and expertise in the field of Accounting, finance, cost accounting, security analysis and portfolio management and management, MS office is at my fingertips, I want my client to take advantage of my practical knowledge. I have been mentoring my client on a freelancer website from last two years, Currently I am working in Telecom company as a financial analyst and before that working as an accountant with Pepsi for one year. I also join a nonprofit organization as a finance assistant to my job duties are making payment to client after tax calculation, I have started my professional career from teaching I was teaching to a master's level student for two years in the evening.
My Expert Service
Financial accounting, Financial management, Cost accounting, Human resource management, Business communication and report writing. Financial accounting : • Journal entries • Financial statements including balance sheet, Profit & Loss account, Cash flow statement • Adjustment entries • Ratio analysis • Accounting concepts • Single entry accounting • Double entry accounting • Bills of exchange • Bank reconciliation statements Cost accounting : • Budgeting • Job order costing • Process costing • Cost of goods sold Financial management : • Capital budgeting • Net Present Value (NPV) • Internal Rate of Return (IRR) • Payback period • Discounted cash flows • Financial analysis • Capital assets pricing model • Simple interest, Compound interest & annuities
4.40+
65+ Reviews
86+ Question Solved
Related Book For 

Question Posted:
Students also viewed these Sciences questions
-
On 06 June 2020, the Government of the National Capital Territory of Delhi (GNCTD) floated a tender for the 'Development of Government Officers Colonies in the NCT of Delhi, and for the Improvement,...
-
Assign R or S configuration to each chirality center in the following monosaccharide?s and tell whether each is a D sugar or an L sugar: (b) (a) - (c) - - - - - - - - - - CH- CH2 CH2
-
The break-even point in sales dollars for Culver Company is $360,000 and the company's contribution margin ratio is 30%. If Culver Company desires an income of $84,000, sales would have to total? A....
-
Should courts ever enforce illegal contracts? If illegal contracts are void as a matter of law, what is the court enforcing? If courts will use equity powers or other roundabout ways to enforce...
-
Who won? Why?
-
Medical Arts Hospital (MAH) uses a hospital-wide overhead rate based on nurse-hours. The intensive care unit (ICU), which has 30 beds, applies overhead using patient-days. Its budgeted cost and...
-
One critical-thinking skill is a heightened awareness of the danger of reaching a conclusion prior to acquiring missing information that were it known would have a reasonable probability of altering...
-
The wheat harvesting season in the American Midwest is short, and most farmers deliver their truckloads of wheat to a giant central storage bin within a two-week span. Because of this, wheat-filled...
-
Define a set X of strings in the symbols 0 and 1 as follows. B. 0 and 1 are in X. R. If x and y are in X, so is xxyy. R2. If x and y are in X, so is xyx. Explain why the string 01001011 EX using the...
-
Suppose a sample of an optically active substance has an observed rotation of 110. The scale on the analyzer of a polarimeter is circular, so 110 is the same as 2350 or 1370. How would you determine...
-
Draw line-and-wedge representations for each of the following chiral molecules. Use models if necessary. (D 5 deuterium = 2 H, a heavy isotope of hydrogen.) Note that several correct structures are...
-
Do satisfied employees provide better customer service? What has been your experience? Do employees that seem happy at work provide better customer service? Discuss.
-
What should you as a solicitor specialising in media law advise your media client to do if it wishes to expand its operations from simply television broadcasting to local radio as well as,...
-
A defendant is believed by many to be guilty of child Which of the following is one of the 3 media stereotypes of professional law enforcement created by the media? of the following is one of the 3...
-
Discuss and interpret the nature of copyright in a world of social media. Consider whether law protects (or should protect) the copyright of individuals on social media. ?
-
Explain how and why social media is being used by law enforcement. Also discuss the problems that may result from the adoption of social media by law enforcement agencies.?
-
Need news article describing a legal issue dealing with media or mediated communication (e.g., the internet, social media, communication law). Summarize the article and indicate which source of law...
-
Draw a supply and demand graph and identify the areas of consumer surplus and producer surplus. Given the demand curve, what impact will an increase in supply have on the amount of consumer surplus...
-
For each of the following reactions, express the equilibrium constant: a) H20 (I) H2 (g) + 02 (g) Ke = 1.0x107 b) Fe2 (g) 2F (g) Ke= 4.9 x 10-21 c) C (s) + O2 (g) d) H2 (g) + C2H4 (g) C2H6 (g) Ke =...
-
Give IUPAC names for the followingstructures: (c) CH (b) (a) " SH (f) CH (e) (d) CHCH0- CHH 5. NO2 SCH3 SCH3 (i) (h) CH CH CH3CH2CHCHCHSCHCH3 CH CH (g) CCH H
-
Predict the products of the following ether cleavagereactions: .CH3 (a) (b) "CH-CH HI CFCO2H CH H20 (d) HI %3DCHH2CH CH CH20-H2CH3 (c) I H20 CH
-
How would you prepare the following ethers? (b) "CH-CH "CH (c) (a) - CH (e) (d) CH "CH H (f) - H
-
Current Attempt in Progress Vermillion Ltd. prepares an aging schedule for its accounts receivable at the end of each month and records credit losses monthly. The following selected information is...
-
You want to be able to withdraw $25,000 each year for 15 years. Your account earns 7% interest. How much do you need in your account at the beginning?
-
Based on historical data, the Hospital of St. Jacques wants to have 7 bags of B positive blood to every 2 bags of B Negative, furthermore, they want to have 3 bags of B negative blood for every 1 bag...

Study smarter with the SolutionInn App


