Outline a preparation of sulfanilamide, a sulfa drug, from aniline and any other reagents. HN- 0=5 O
Question:
Outline a preparation of sulfanilamide, a sulfa drug, from aniline and any other reagents.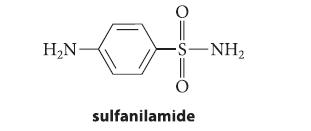
Transcribed Image Text:
H₂N- 0=5 O -S—NH, sulfanilamide
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 100% (QA)
HN Use an acetyl protecting group as outlined CHCCI pyridine CHCNH ...View the full answer

Answered By

ALBANUS MUTUKU
If you are looking for exceptional academic and non-academic work feel free to consider my expertise and you will not regret. I have enough experience working in the freelancing industry hence the unmistakable quality service delivery
4.70+
178+ Reviews
335+ Question Solved
Related Book For 

Question Posted:
Students also viewed these Sciences questions
-
Outline a Preparation of the following compounds from aniline and any other reagents. Sulfathiazole, a sulfa drug. H2N sulfathiazole
-
Outline a preparation of each of the following compounds from aniline and any other reagents. (a) 2,4-dinitroaniline (b) Sulfathiazole, a sulfa drug HN- -NH- sulfathiazole HN- 2-aminothiazole
-
Outline syntheses of the following compouods ftorn the indicated starting materials. (a) p-iodoaaisole from phenol and any other reagents (b) n-bromoiodobenzene frorn nitobenzene
-
3. Complete all parts. Let f(x) = I 2+1 on the interval [0, 2). = x 2+1 (a) Explain why the function f(x) on interval [0,2] satisfies the Mean Value Theorem. (b) find all numbers c that satisfy the...
-
What are the main functions of an accounting clerk?
-
Some businesses extend purchasing department employees the authority to perform a broader range of activities than do other businesses. What activities do all purchasing departments perform? What...
-
Reconsider the data from Problem 56. What is the capital recovery cost of Model 334A? Data from problem 56 Octavia Bakery is planning to purchase one of two ovens. The expected cash flows for each...
-
Consider the following network for conducting a two week (10 working days) computer training class: (a) Construct a schedule showing: ESs for all activities LSs for all activities Slacks for all...
-
(30 Pts.) Convert the following for loop into a while loop n=[] For Loop for x in range(100,0,-1): if x%10==0: n.append(x) print(n) While Loop (Write your answer below)
-
As shown in the following equation, when (R)-1-deuterio-1-butanamine is diazotized with nitrous acid in water, the alcohol product formed has the S configuration (D = 2 H). (a) Give the...
-
Provide a reaction mechanism for the reaction shown in Eq. 23.36. NH 5- + 3 Br Br NH Br Br + 3HBr (23.36)
-
Donated medicines that normally would be purchased by a government hospital should be recorded at fair market value and should be credited directly to a. other operating revenue. b. nonoperating...
-
Below is a partial Statement of Financial Position of Malika Bhd as at 31 December 2021: Malika Bhd Partial Statement of Financial Position As at 31 December 2021 i. ii. As at 31 December 2021,...
-
Write a prolog program that will determine the equal integers in two different sorted lists and will form a third list that will consist of these equal integers. The predicate that you will implement...
-
An investment advisor has presented you with the following proposal: Purchase an investment with an immediate cash payment and then expect to receive a $20 payment in one year. The payments are...
-
C The price of product X is reduced from $45 to $20 and, as a result, the quantity demanded increases from 20 to 25 units. From this we can conclude that the demand for X in this price range: O is...
-
The North American Free Trade Agreement (NAFTA) was a regional trade agreement that included A. five countries in Central America. B. Mexico, the United States, and Canada. C. North and South America.
-
Compare and contrast BPA, BPI, and BPR. Which is most risky? Which has the greatest potential value?
-
A routine activity such as pumping gasoline can be related to many of the concepts studied in this text. Suppose that premium unleaded costs $3.75 per gal. Work Exercises in order. Use the...
-
The phenomenon of configuration inversion in a chemical reaction was discovered in 1896 by Paul Walden (Section 6.6). Walden's proof of configuration inversion was based on the following cycle: (a)...
-
(R)-(3-Chloro-2-methylpropyl) methyl ether (A) on reaction with azide ion (N3-) in aqueous ethanol gives (S)-(3-azido-2-methylpropyl) methyl ether (B). Compound A has the structure...
-
Predict the structure of the product of this reaction: The product has no infrared absorption in the 1620-1680-cm-1 region. NaOH in aqueous EtOH CI HS
-
Data table Estimated Estimated manufacturing overhead costs quantity of allocation Predetermined base overhead rate Direct labor hours $ 1,500,000 60,000 $25.00 per DL hour Direct labor dollars $...
-
Perpetual Inventory Using FIFO Beginning inventory, purchases, and sales for Item Zeta9 are as follows: Oct. 1 Inventory 51 units @ $25 7 Sale 34 units 15 Purchase 57 units @ $29 24 Sale 22 units...
-
Sandra Jones intends to retire in 20 years at the age of 65. As yet, she has not provided for retirement income, and she wants to set up a periodic savings plan to do this. She has the opportunity to...

Study smarter with the SolutionInn App


