Outline a synthesis of each of the following compounds from either diethyl malonate or ethyl acetate. Because
Question:
Outline a synthesis of each of the following compounds from either diethyl malonate or ethyl acetate. Because the branched amide bases are relatively expensive, you may use them in only one reaction.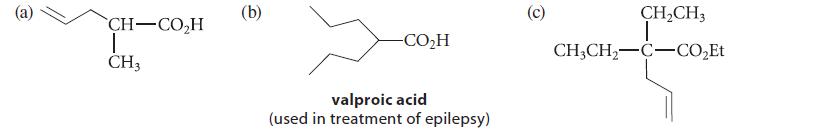
Transcribed Image Text:
CH–COH T CH3 (b) -CO,H valproic acid (used in treatment of epilepsy) (c) CH₂CH3 T CH3CH₂-C-CO₂Et
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 66% (3 reviews)
Because part c involves formation of a carboxylic acid with a quaternary acarbon and because ...View the full answer

Answered By

Arshad Ahmad
Well, I am really new to tutoring but I truly believe a good student can be a better teacher. I have always been a topper at school. I passed my Chartered Accountancy at a very young age of 23, a rare feat for most of the students. I am really dedicated to whatever work I do and I am very strict regarding deadlines. i am always committed and dedicated to whatever work allotted to me and I make sure it is completed well within deadline and also I try to give my best in whatever I do. Hope we will have a good time studying together.
5.00+
1+ Reviews
10+ Question Solved
Related Book For 

Question Posted:
Students also viewed these Sciences questions
-
Outline a synthesis of each of the following compounds from the indicated strating material and any other reagents. (a) N-(sec-buty1) -N-ethylaniline from chlorobenxene (b) (c) (CH,),C from phenol NH...
-
Outline a synthesis of each of the following compounds from a -keto ester; then show how the -keto ester itself can be prepared. CHj
-
Outline a synthesis of each of the following compounds from the indicated starting materials and any other reagents. (a) 1-cyclohexyl-2-methyl-2-prupanol from bromocyclohexane (b) PhNHCH2CH2CH(CH3)2...
-
Misty Cumbie worked as a waitress at the Vita Caf in Portland, Oregon. The caf was owned and operated by Woody Woo, Inc. Woody Woo paid its servers an hourly wage that was higher than the states...
-
California Pool Supplies inventory data for the year ended December 31, 2012, follow: Assume that the ending inventory was accidentally overstated by $2,400. Requirement 1. What are the correct...
-
What is a bond indenture?
-
Are the input specifiers for scanf() identical to the output specifiers for printf()?
-
Jackplum Vineyards, whose fiscal year begins on November 1, has just completed a record-breaking year producing and selling wine. Its inventory account balances on October 31 of this year were...
-
The scope of a project is expanded through the formal change control process, causing the schedule to be extended beyond the original due date. This is an example of which type of influence?
-
Predict the product formed when the conjugate-base enolate ion of ethyl 2-methylpro panoate (shown in Eq. 22.81) is treated with bromobenzene and a catalytic amount of Pd[P(t-Bu) 3 ] 4 , and explain...
-
Fatty acids are degraded to acetyl-CoA in fatty-acid metabolism. The enzyme that catalyzes this conversion, acyl-CoA acetyl transferase, contains a nucleophilic thiol group in its active site and...
-
Find the points of intersection of the graphs of the equations. x - y = -5 x - y = 1
-
Cesium-137 is radioactive and has a half life of 30. years. Calculate the activity of a 1.4 mg sample of cesium-137. Give your answer in becquerels and in curies. Be sure your answer has the correct...
-
m m a car travels at 12- for 15s. It then speeds up with a constant acceleration of 1.0 for 3.0s. at the end S S what is its velocity?
-
Could you elucidate the significance of information technology within the supply chain, delineating its multifaceted role and impact on operational efficiency and strategic decision-making processes?
-
A candle is sitting in front of a concave mirror at a distance of 1 2 . 2 cm . It is found that an image of the candle appears at 2 5 . 0 cm . What is the focal length of the mirror?
-
Could you elaborate on the concept of segmentation within the realm of strategic supply chain management, detailing its significance and implications for optimizing operational performance and...
-
Define the term manufacturing cycle efficiency.
-
What mass of KBr (in grams) should you use to make 350.0 mL of a 1.30 M KBr solution?
-
(a) What monobromo allylic substitution products would result from reaction of each of the following compounds with NBS in the presence of peroxides and/or light? (b) In the case of isomeric products...
-
Benzylic radicals, due to the adjacent benzene ring, have even greater possibility for de-localization than allylic radicals. Draw contributing resonance structures that show this delocalization for...
-
When propylbenzene reacts with chlorine in the presence of UV radiation, the major product is 1-chloro-1-phenylpropane. Both 2-chloro-1-phenylpropane and 3-chloro- 1-phenylpropane are minor products....
-
What approach is found frequently among companies with strong engineering orientations, pharmaceutical and biomedical firms, financial services, and many high technology companies?
-
A company claims that it has valuable resources. How might another firm determine whether their claim is accurate? determine whether the corporation attracts highly educated employees determine...
-
t Sales Contribution margin Fixed costs Skittles Company $ 6,485,950 4,585,950 3,519,450 Starburst Company $ 4,218,000 1,443,000 888,000 (1) Compute the degree of operating leverage (DOL) for each...

Study smarter with the SolutionInn App


