Predict max for the UV absorption of each of the following compounds. (a) Et H T
Question:
Predict λmax for the UV absorption of each of the following compounds.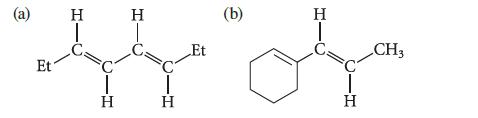
Transcribed Image Text:
(a) Et H T H H T H Et (b) H T H CH3
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 100% (1 review)
Based on the image you sent here are my predictions for the max values for the UV absorption of each ...View the full answer

Answered By

Salmon ouma
I am a graduate of Maseno University, I graduated with a second class honors upper division in Business administration. I have assisted many students with their academic work during my years of tutoring. That has helped me build my experience as an academic writer. I am happy to tell you that many students have benefited from my work as a writer since my work is perfect, precise, and always submitted in due time. I am able to work under very minimal or no supervision at all and be able to beat deadlines.
I have high knowledge of essay writing skills. I am also well conversant with formatting styles such as Harvard, APA, MLA, and Chicago. All that combined with my knowledge in methods of data analysis such as regression analysis, hypothesis analysis, inductive approach, and deductive approach have enabled me to assist several college and university students across the world with their academic work such as essays, thesis writing, term paper, research project, and dissertation. I have managed to help students get their work done in good time due to my dedication to writing.
5.00+
4+ Reviews
16+ Question Solved
Related Book For 

Question Posted:
Students also viewed these Sciences questions
-
Predict the products when each of the following compounds is treated with NBS and irradiated with UV light: (a) (b) (c) (d)
-
Sunscreen contains compounds that absorb ultraviolet light. When sunscreen is applied to skin, it prevents ultraviolet light from reaching the skin. The graph that follows shows the absorbance of...
-
The UV spectrum of 1-phenylprop-2-en-1-ol shows an intense absorption at 220 nm and a weaker absorption at 258 nm. When this compound is treated with dilute sulfuric acid, it rearranges to an isomer...
-
If a company is very diversified, (a) it makes it easier to classify the company by industry. (b) it would not be necessary to provide any segmented information. (c) it can limit the usefulness of...
-
What type of transaction is a cash payment to creditors? How does this type of transaction affect the accounting equation?
-
Shanken Corp. issued a 30-year, 6.2 percent semiannual bond 7 years ago. The bond currently sells for 108 percent of its face value. The companys tax rate is 35 percent. a. What is the pretax cost of...
-
Panther Racing, LLC, is involved in competitive racing, including the development, maintenance, and operation of a racing team in the IndyCar series. Beginning in 2008, the National Guard annually...
-
Entries for Held-to-Maturity Securities on January 1, 2010, Jennings Company purchased at par 10% bonds having a maturity value of $300,000. They are dated January 1, 2010, and mature January 1,...
-
Breast cancer patients in a London, England, hospital were being treated for spinal metastases. They were followed over a five-year period, with their ambulatory state being recorded before treatment...
-
Refer to Fig. 15.11. At what pH does the fluorescence of fluorescein have the greater quantum yield, pH = 9 or pH = 5? How do you know? relative absorbance 300 488 nm pH=9 pH = 5 400 wavelength, nm...
-
(a) From the extinction coefficient of isoprene (10,750 M 1 cm 1 ) and its observed absorbance at 222.5 nm (Fig. 15.5), calculate the concentration of isoprene in mol L 1 (assume a 1 cm light path)....
-
Why is it necessary to raise the temperature of P/M compacts slowly to the temperature of sintering?
-
What are the four economic types of mergers?
-
What is breakup value?
-
A decrease in demand while supply remains unchanged will lead to ______. a) an increase in equilibrium price and equilibrium quantity b) a decrease in equilibrium price and equilibrium quantity c) an...
-
What is an action standard? How might defining action standards lead to better results?
-
Explain how a company can use the futures market to hedge against rising raw materials prices.
-
Don is the production manager for Corporate Manufacturing Facilities (CMF). CMF works three production shifts per day. Because Don is so integral to CMF's operations, the company requires him to live...
-
Provide an example of an aggressive accounting practice. Why is this practice aggressive?
-
How many 13C NMR absorptions would you expect for cis-1, 3-dimethyl- cyclohexane for trails-1, 3-dirnethylcyclohexane? Explain.
-
Assume that you have a compound with formula C 3 H 6 O. (a) How many double bonds arid/or rings does your compound contain? (b) Propose as many structures as you can that fit the molecular formula....
-
How could you use 1H and 13C NMR to help you distinguish among the following isomeric compounds of formulaC4H8? CH2-CH2 H2H CH CCH3HCH CH2-CH2 CHH2
-
what is investment banking & investment bank ?
-
describe the function of global investment banking ? 2)how you will utilize your academic skills and experience to add value to investment banking division?
-
Discuss the exposure of investment banking in Saudi Arabia. After reading the bellow link: https://www.hsbcsaudi.com/en/Investment/Investment-Banking-Advisory

Study smarter with the SolutionInn App


