Refer to Fig. 15.11. At what pH does the fluorescence of fluorescein have the greater quantum yield,
Question:
Refer to Fig. 15.11. At what pH does the fluorescence of fluorescein have the greater quantum yield, pH = 9 or pH = 5? How do you know?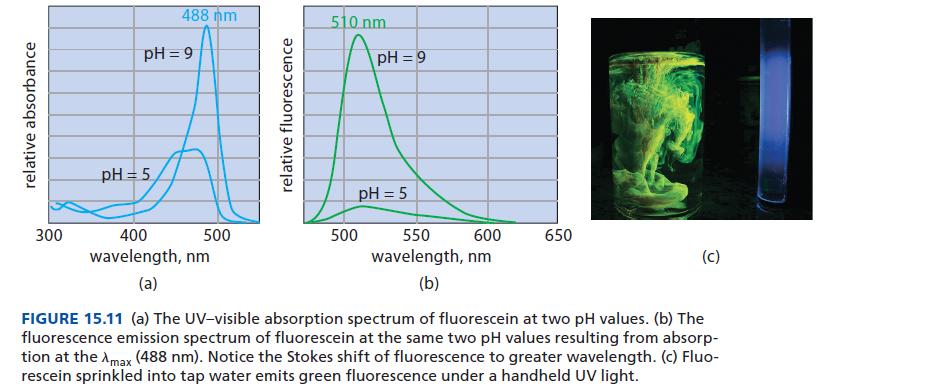
Transcribed Image Text:
relative absorbance 300 488 nm pH=9 pH = 5 400 wavelength, nm (a) 500 relative fluorescence 510 nm pH=9 pH = 5 500 600 550 wavelength, nm (b) 650 (C) FIGURE 15.11 (a) The UV-visible absorption spectrum of fluorescein at two pH values. (b) The fluorescence emission spectrum of fluorescein at the same two pH values resulting from absorp- tion at the Amax (488 nm). Notice the Stokes shift of fluorescence to greater wavelength. (c) Fluo- rescein sprinkled into tap water emits green fluorescence under a handheld UV light.
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 100% (1 review)
The figure presented shows the UVvisible absorption spectrum and the fluorescence emission spectrum ...View the full answer

Answered By

Nyron Beeput
I am an active educator and professional tutor with substantial experience in Biology and General Science. The past two years I have been tutoring online intensively with high school and college students. I have been teaching for four years and this experience has helped me to hone skills such as patience, dedication and flexibility. I work at the pace of my students and ensure that they understand.
My method of using real life examples that my students can relate to has helped them grasp concepts more readily. I also help students learn how to apply their knowledge and they appreciate that very much.
4.00+
1+ Reviews
10+ Question Solved
Related Book For 

Question Posted:
Students also viewed these Sciences questions
-
You will need to save money to reach your financial goals. To save money, you will need a plan to show what money comes in and what money goes out. This is known as a budget. You may already have a...
-
You are interviewing a candidate for a position at a call center. You need someone polite, courteous, patient, and dependable. The candidate you are talking to seems nice. But how do you know who is...
-
The following question was asked of executives: How do you know when to cut off research? The answers given: Thats a good question, a very good question, and some people dont know when to cut it off....
-
A tape player has three operations: play, fast forward and fast play. Play and fast forward are activated using the play and fast forward button respectively. These operations can be cancelled using...
-
When are expenses recognized under accrual accounting?
-
Dawn Browne, an investment broker, has been approached by client Jack Thomas about the risk of his investments. Dawn has recently read several articles concerning the risk factors that can...
-
Many single women and married couples use donated sperm to conceive children each year. Pennsylvania resident Donna Donovan decided to use donated sperm from Idant Laboratories, a New York sperm bank...
-
Sutton Pointers Corporation expects to begin operations on January 1, 2015; it will operate as a specialty sales company that sells laser pointers over the Internet. Sutton expects sales in January...
-
please explain this answer by Relational model drawing Question # 01: Convert the given ER model into Relational Model Fname Minit Lname Bdate Name Address Salary Sen Sex Locations WORKS FOR (4,N)...
-
One of the following compounds has an intense yellow fluorescence when irradiated with UV light. Which one do you think it is, and why? HC. CH3 0=S=O A NH HC. N CH 0=S=0 NH B HC. CH3 0=S=0 NH C H3C....
-
Predict max for the UV absorption of each of the following compounds. (a) Et H T H H T H Et (b) H T H CH3
-
A market failure is said to occur ______. a. When costs are imposed on some people without their consent b. When the market economy fails to allocate resources efficiently c. When one party prevents...
-
Identify what you believe are the three most important key performance indicators (KPIs) that the seafood restaurant Red Lobster should use to measure its success. Explain.
-
What is the difference between an operating merger and a financial merger?
-
Should preferred stock be considered as equity or debt? Explain.
-
What are some reasons for the current merger wave?
-
Give an example of an impediment to problem definition.
-
Determine whether the taxpayer has received taxable income in each of the following situations. Explain why any amount(s) may be excluded: a. Jim is an employee of Fast Tax Prep, Inc. All employees...
-
Explain the regulation of the secretions of the small intestine.
-
The acid-catalyzed dehydration of 1-methylcyclohexanol yields a mixture of two alkenes. How could you use 1H NMR to help you decide which waswhich? CH CH - 0* CH2
-
How could you use 1H NMR to distinguish between the following pairs ofisomers? (a) CH3CH=CHCH2CH3 and CH2 H H2H () CH20CH2CH and CH3OCH2CH2CH3 (c) CHC,H and CH3CH2CH3 (d) HCICH)H and CH3CH=CHCH3
-
Propose structures for compounds with the following formulas that show only one peak in their 1H NMR spectra: (a) C5H12 (b) C5H10 (c) C4H8O2
-
help me with technology innovation in finance and investment banking. Need innovation done in finance , could be retail, commercial and investment banking. Need to write an assignment so pointers or...
-
What is the molecular structure and causative agent behind the formation of a thymine dimer in DNA?
-
A broker-dealer must establish information barriers between which two departments? give reasons

Study smarter with the SolutionInn App


