Propose a curved-arrow mechanism for each of the reactions given in Fig. P17.46. (b) CH3(CH)3 C=C-CH-Br +
Question:
Propose a curved-arrow mechanism for each of the reactions given in Fig. P17.46.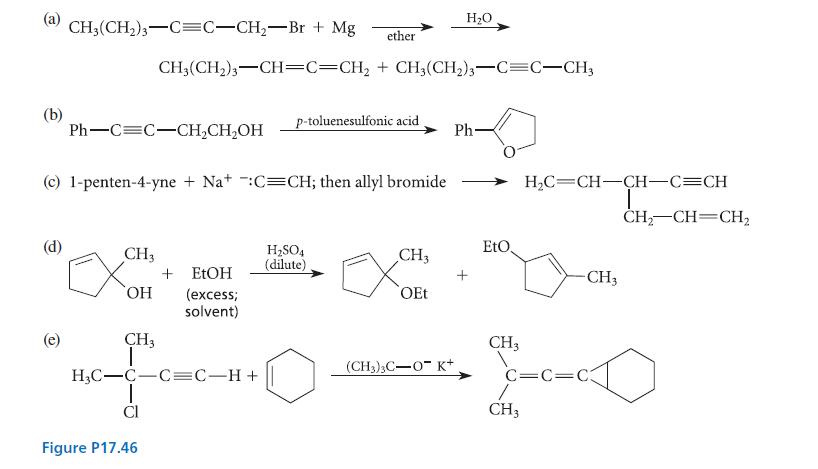
Transcribed Image Text:
(b) CH3(CH₂)3 C=C-CH₂-Br + Mg (e) Ph-C=C-CH₂CH₂OH CH3 (c) 1-penten-4-yne + Na+ :C=CH; then allyl bromide + EtOH OH (excess; solvent) CH3 CH₂(CH₂)3-CH=C=CH₂ + CH3(CH2)3-C=C-CH3 H₂C-C-C=C-H+ Cl Figure P17.46 ether p-toluenesulfonic acid. H₂SO4 (dilute) CH3 OEt H₂O (CH3)3C-OK+ Ph- EtO. CH3 H₂C CH-CH-C=CH L CH3 -CH3 C=C=C3 CH₂ CH CH₂
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 100% (2 reviews)
a Propargylic Grignard reagents like allylic Grignard reagents are an equilibrium mixture of two con...View the full answer

Answered By

Niala Orodi
I am a competent and an experienced writer with impeccable research and analytical skills. I am capable of producing quality content promptly. My core specialty includes health and medical sciences, but I can competently handle a vast majority of disciplines.
5.00+
1+ Reviews
10+ Question Solved
Related Book For 

Question Posted:
Students also viewed these Sciences questions
-
The total amount paid in by__________for the shares they purchase is described as common stock. Select answer from the options below creditors bondholders employees stockholders
-
Give a curved-arrow mechanism for each of the reactions given in Fig. P20.51. OC,H, dil. HCI (catal ,- OC2Hs an orthoester 0 CIH O C CH dil HOI (catalyst) CH OH (e) H,C ,--, carbon monoxide CH Ph Ph...
-
Give a curved-arrow mechanism for each of the reactions given in Fig. P20.52. (a) (b) OEt 1 H3C-C-OEt + HO T OEt an orthoester (c) HC H3C C-CH3 COH dil. HCl (catalyst) CH3OH + C=CH + :C=O: dil. HCI...
-
Modify BST to add a method rangeSearch () that takes two keys as arguments and returns an iterable over all keys that are between the two given keys. The running time should be proportional to the...
-
An interstellar spaceship travels from the earth to a distant star system 12 light-years away (as measured in the earths frame). The trip takes 15 years as measured on the ship. (a) What is the speed...
-
On December 31, 2012, Cathy Chen, a self-employed certified public accountant (CPA), completed her first full year in business. During the year, she billed $360,000 for her accounting services. She...
-
Using the data in Figure 13.37 for the venules, and assuming a typical blood flow rate of 5.0 L per minute, a. What is the flow speed in the venules? b. What is the pressure difference across a 1.0...
-
You are auditing the financial statements for your new client, Paper Pack-aging Corporation, a manufacturer of paper containers, for the year ended March 31, 2014. Paper Packagings previous auditors...
-
Vaughan Trophies had beginning inventory of 15,000 units in Department #1. During the period they started 64,000 units. At the end of the period there were 30,000 units in ending WIP that were 40%...
-
Specify the relationship(s) of the compounds in each of the following sets. Choose among the following terms: identical compounds, epimers, anomers, enantiomers, diastereomers, constitutional...
-
Consider the relative rates of the two solvolysis reactions in acetic acid solvent shown in Fig. P17.50. (a) Suggest a reason that compound A undergoes solvolysis much faster than compound B. (b)...
-
What do you understand by cost-plus contract and Escalation clause in contract costing?
-
Discuss the advantages and disadvantages of ratio analysis. Montgomery Organizations plc, whose annual accounts are given below. The firm has 14,685,856 shares and the share price is 2.78. Fixed...
-
Calculating Future Values Calculate the future value of a 100 cash flow for the following combinations of rates and times: (a) r = 8%; t = 10 years (b) r = 8%; t = 20 years (c) r = 4%; t = 10 years...
-
When Devon experiences an increase in wealth, her total utility increases as depicted in the accompanying table. a. Graph Devons utility function. Does it exhibit diminishing marginal utility? b. Her...
-
Interest You work for a jewellers and have sourced a good goldsmith who is able to sell you 100 ounces of gold for 100,000. You approach your two main customers. Mr Noel says he will buy the gold...
-
If Apollos functional currency is the FC, which of the following statements holds? (a) A translation gain of $480,000 is recorded in income. (b) A translation loss of $480,000 is taken to Foreign...
-
The exponential distribution can be used to solve Poisson-type problems in which the intervals are not time. The Airline Quality Rating Study published by the U.S. Department of Transportation...
-
Drainee purchases direct materials each month. Its payment history shows that 65% is paid in the month of purchase with the remaining balance paid the month after purchase. Prepare a cash payment...
-
Without drawing the MOs, state whether the 7r-molecular orbital 6 in 1,3,5,7,9-decapentaene (a 10-carbon conjugated alkene) is symmetric or anti symmetric with respect to the reference plane; is...
-
What do the pericyclic selection rules have to say about the position of equilibrium in each of the reactions given in Fig. P27.30? Which side of each equilibrium is favored and why? Fig. P27.30 (a)...
-
What stereoisomer of A also gives compound C on heating?
-
Investment X offers to pay you $CF per year for eight years, starting one year from now, in return for your investment today (PV or CFO) of $35,788. What is $CF if the interest rate for this project...
-
Jack and Diane's have received one more offer for a home mortgage. This option - Option C, is described below. The purchase cost ($212,400) of the condo will remain the same and the time they expect...
-
How does the implementation of Kaizen philosophy contribute to the perpetuation of continuous improvement within complex organizational structures?

Study smarter with the SolutionInn App


