Rank the following compounds in order of increasing acidity. Explain your answers. OH A OCH3 OH +
Question:
Rank the following compounds in order of increasing acidity. Explain your answers.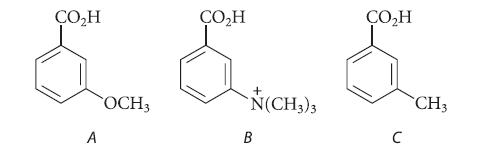
Transcribed Image Text:
ÇO₂H A OCH3 ÇO₂H + N(CH3)3 B CO₂H C CH3
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 100% (3 reviews)
The trimethylammonium substituent in B has the greatest acidstrengthening polar ...View the full answer

Answered By

PALASH JHANWAR
I am a Chartered Accountant with AIR 45 in CA - IPCC. I am a Merit Holder ( B.Com ). The following is my educational details.
PLEASE ACCESS MY RESUME FROM THE FOLLOWING LINK: https://drive.google.com/file/d/1hYR1uch-ff6MRC_cDB07K6VqY9kQ3SFL/view?usp=sharing
3.80+
3+ Reviews
10+ Question Solved
Related Book For 

Question Posted:
Students also viewed these Sciences questions
-
Rank the following compounds in order of increasing reactivity (least reactive first) in an SN1 solvolysis reaction in aqueous acetone. Explain your answers. (The structure of tert-cumyl chloride is...
-
Rank the following compounds in order of increasing reactivity in bromination. In each case, indicate whether the principal monobromination products will be the ortho and para isomers or the meta...
-
Shock Electronics sells portable heaters for $35 per unit, and the variable cost to produce them is $22. Mr. Amps estimates that the fixed costs are $97,500. a. Compute the break-even point in units....
-
Suppose a Bubba store purchases $61,000 of womens sportswear on account from Tomas on July 1, 2012. Credit terms are 2/10, net 45. Bubba pays electronically, and Tomas receives the money on July 10,...
-
A mutual fund plans to purchase $ 10 million of 20-year T-bonds in two months. The bonds are yielding 7.68 percent. These bonds have a duration of 11 years. The mutual fund is concerned about...
-
Can you present a graphic that presents the payroll disbursement amounts by date for the contact employee who has been terminated but has been paid after termination (i.e., ghost employees)?
-
Union Express has 60 tons of cargo that needs to be shipped from Boston to Dallas. The shipping capacity on each of the routes Union Express planes fly each night is shown in the following table:...
-
You are director of HRM (international) for an international firm operating mainly in Africa. The business strategy is to expand operations into the Far East, starting in Malaysia. A number of...
-
Explain why all efforts to synthesize a carboxylic acid containing the isotope oxygen-18 at only the carbonyl oxygen fail and yield instead a carboxylic acid in which the labeled oxygen is...
-
Draw the structures and give the names of all the dicarboxylic acids with the formula C 6 H 10 O 4 . Indicate which are chiral, which would readily form cyclic anhydrides on heating, and which would...
-
What does it mean to be socialized into the culture of an organization? How might we understand technical documents as socialization tools?
-
A country can produce either 116 units of good Y or 210 units of good X. If they want to produce 36 units of good Y, how many units of good X are they able to produce?
-
Calculate and sign the tax multiplier (dY/dt) in the following model. Y = C[(1-t)Y] + I[ i ] + G M = L[i, (1-t)Y]
-
c) List some of the types of resource that a typical IS project needs. (4 marks.) For FOUR (4) of your chosen resource types, summarise how the resource is normally obtained and managed. (4 marks.)
-
a firm produces output according to the production function Q=4K+8L where K is capital and L is labour. in this production function are capital and labour (a) perfect complements (b) perfect...
-
When you are looking for a job, you act as a supplier (of labor) and companies stand as your buyer. Would you work for a company with a price ceiling for labor? Justify your answer. Write the answer...
-
Scrooge and Zilch, a public accounting firm in London, is engaged in the preparation of income tax returns for individuals. The firm uses the weighted-average method of process costing for internal...
-
Hotel Majestic is interested in estimating fixed and variable costs so that the company can make more accurate projections of costs and profit. The hotel is in a resort area that is particularly busy...
-
Although Hückel's rule (Section 14.7) strictly applies only to monocyclic compounds, it does appear to have application to certain bicyclic compounds, if one assumes use of resonance...
-
(a) In 1960 T. Katz (Columbia University) showed that cyclooctatetraene adds two electrons when treated with potassium metal and forms a stable, planar dianion, C8H82- (as the dipotassium salt): Use...
-
Although none of the [10]annulenes given in Section 14.7B is aromatic, the following 10 p-electron system is aromatic: What factor makes this possible?
-
Problem 6: Rubber Band Redux The potential energy of a rubber band of relaxed length L when it is stretched to a length may be shown to be l U -k = 1 k ( 1 - 31 + 21 ). (3.60) where k is a constant....
-
Consider the expression 1 2z 1 1+z 1 - d x d y d = + L L L de dy dz I = L dy 0 2z (a) Rewrite I using the order of integration dy dx dz. (b) Using the result from part (a) (or otherwise), evaluate I....
-
Question 3. Prove by induction the identity (n+1)(n+2)... (2n-1) 2n 1.3.5.... (2n-1) for any natural n. 2n

Study smarter with the SolutionInn App


