State whether each of the following molecules is achiral or chiral. Cl H-C-Br -EL F (b) CI
Question:
State whether each of the following molecules is achiral or chiral.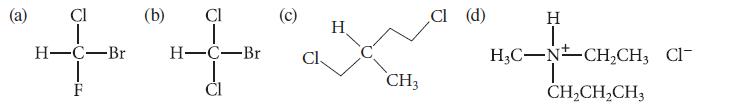
Transcribed Image Text:
Cl H-C-Br -EL F (b) CI H-C-Br Cl H CH3 Cl (d) H I H₂C-NCH₂CH, Cl- T CH₂CH₂CH3
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 100% (3 reviews)
a This compound is chiral b This compound is achiral c Thi...View the full answer

Answered By

WAHIDUL HAQUE
hello,
I'm a professional academic solution provider working as a freelance academic solution provider since 7 years. I have completed numerous projects. Help lots of students to get good marks in their exams and quizzes. I can provide any type of academic help to your homework, classwork etc, if you are a student of Accounting, Finance, Economics, Statistics. I believe in satisfying client by my work quality, rather than making one-time profit. I charge reasonable so that we make good long term relationship. why will you choose me? i am an extremely passionate, boldly honest, ethically driven and pro-active contractor that holds each of my clients in high regards throughout all my business relations. in addition, I'll always make sure that I'm giving my 100% better in every work that will be entrusted to me to be able to produce an outcome that will meet my client's standards. so if you are a student that is now reading my profile and considering me for your academic help. please feel free to look through my working history, feedback and contact me if you see or read something that interests you. I appreciate your time and consideration.
regards
4.90+
233+ Reviews
368+ Question Solved
Related Book For 

Question Posted:
Students also viewed these Sciences questions
-
Indicate whether each of the following statements is true or false. If false, explain why. (a) In some cases, constitutional isomers are chiral. (b) In every case, a pair of enantiomers have a mirror...
-
Indicate whether each of the following statements is true or false. If false, explain why. (a) In every case, pair of enantiomers have a mirror- image relationship. (b) If a compound has an...
-
Identify whether each of the following compounds is chiral or achiral: a. b. c. d. e. f. g. h. i. j. k. l. m. n. o. p. CI
-
The 10-year Coupon Bond has a face value of $1,000, the annual coupon rate is 5 percent (out of its face value), the yield to maturity is 10 percent. (2.a) show me the cash flows of this coupon bond,...
-
Could a plaintiff win such a case without showing that she had contracted the HIV virus?
-
Choose the correct answer for each of the following questions. 1. On November 15, 20X3, Chow Inc., a U.S. company, ordered merchandise FOB shipping point from a German company for ¬200,000. The...
-
Discuss the main recognition and measurement issues that must be considered when preparing the income statement.
-
The following selected transactions relate to investment activities of Ornamental Insulation Corporation during 2018. The company buys debt securities, intending to profit from short-term differences...
-
9 A monkey is swinging from a tree. On the first swing, she passes through an arc of 10 m. With each swing, she passes through an arc of the previous swing. the length 10 What is the total distance...
-
Determine the stereochemical configuration of the following enantiomer of 3-chloro-1-pentene: HC CH Cl I CH CHCH3
-
Identify the asymmetric carbon(s) in 4-methyloctane: CH3 CH3CHCHCHCHCHCHCH3 4-methyloctane
-
Working on summer vacation. Recall (Exercise 3.13, p. 145) that a Harris Interactive (July 2013) poll found that 22% of U.S. adults do not work at all while on summer vacation. In a random sample of...
-
What's organizational structure? Also, how does the organizational structure affect internal politics within the firm?
-
Noelle, the owner of all of the shares of ClockCo, an S corporation, transfers her stock to Grayson on April 1. ClockCo reports a $165,000 NOL for the entire tax year, but only $24,750 of the loss...
-
1. Consider the equation ze + sin(ry) + y - In(2)-0. Find the value of dy/dr at the point (0, in 2). 2. Consider the equation sin(x + y) + sin(x+2)+ sin(y+ 2) = 0. Find the values of 8z/0x and Oz/0y...
-
Alphabet Company, which uses the periodic inventory method, purchases different letters for resale. Alphabet had no beginning inventory. It purchased A thru G in January at $ 5 . 0 0 per letter. In...
-
A painting with a mass of 16 kg is suspended by two wires from hooks on a ceiling that connect at a single point on the painting. If the wires have lengths of 18 cm and 24 cm and the distance between...
-
A survey of adults aged 18 and older conducted by Princess Cruises asked how many days into your vacation does it take until you feel truly relaxed (USA today, August 24, 2011). The responses were as...
-
Imagine you are the HR manager at a company, and a female employee came to you upset because she felt a male coworker was creating a hostile work environment by repeatedly asking her out on dates...
-
Treatment of 1, 1-diphenyl- 1, 2-epoxyethane with aqueous acid yields diphenylacetaldehyde as the major product. Propose a mechanism for the reaction. Ph H30+ PHCHCH Ph Ph
-
How would you prepare o-hydroxyphenyl-acetaldehyde from phenol? More than one step is required. HO o-Hydroxyphenylacetaldehyde CH-CO
-
Imagine that you have treated (2R, 3R)-2, 3-epoxy-3-methylpentane with aqueous acid to carry out a ring-opening reaction. (a) Draw the epoxide, showing stereochemistry. (b) Draw and name the product,...
-
A cyclic steam power plant is to be designed for a steam temperature at turbine inlet of 3 5 0 \ deg C and an exhaust pressure of 0 . 8 bar. After isentropic expansion of steam in the turbine, the...
-
Air enters the compressor ( pressure ratio 1 0 ) and the ideal Brayton cycle turbine at 1 6 . 8 5 \ deg C and 8 2 6 . 8 5 \ deg C respectively. Specific heats change with temperature. Draw the...
-
The Airbus 3 1 8 with two Pw 3 6 0 0 engines has the following performance characteristics: Engine: - Dual - spool high 0 bypass turbofan jet. - Fan diameter = 1 . 4 4 m - T = 4 0 kN / engine -...

Study smarter with the SolutionInn App


