Suggest an alkene metathesis reaction that would yield each of the following compounds as a major product.
Question:
Suggest an alkene metathesis reaction that would yield each of the following compounds as a major product.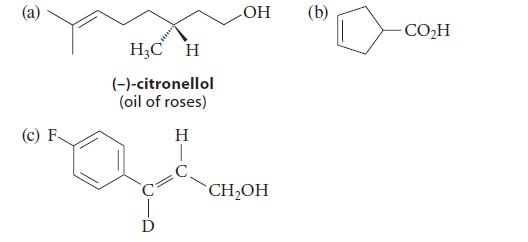
Transcribed Image Text:
(c) F H₂C H (-)-citronellol (oil of roses) H D OH CH₂OH (b) CO₂H
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 0% (1 review)
a b HC H citronellol oil of roses OH ...View the full answer

Answered By

Sheikh Muhammad Ibrahim
During the course of my study, I have worked as a private tutor. I have taught Maths and Physics to O'Level and A'Level students, as well as I have also taught basic engineering courses to my juniors in the university. Engineering intrigues me alot because it a world full of ideas. I have passionately taught students and this made me learn alot. Teaching algebra and basic calculus, from the very basics of it made me very patient. Therefore, I know many tricks to make your work easier for you. I believe that every student has a potential to work himself. I am just here to polish your skills. I am a bright student in my university. My juniors are always happy from me because I help in their assignments and they are never late.
4.90+
14+ Reviews
24+ Question Solved
Related Book For 

Question Posted:
Students also viewed these Sciences questions
-
In each case, give the structure of an eight-carbon alkene that would yield each of the following compounds (and no others) after treatment with ozone followed by dimethyl sulfide. (b)...
-
Each of the following compounds is an aromatic compound bearing a substituent that we did not discuss in this chapter. Using the principles that we discussed in this chapter, predict the major...
-
Write structural formulas for all the alkene products that could reasonably be formed from each of the following compounds under the indicated reaction conditions. Where more than one alkene is...
-
Consider two polluting firms with cost functions C j (x j ,e j ). The inverse demand function is P(X). Firms engage in negotiations about the permit allocation and a transfer price in a first stage...
-
What is the separation distance between clocks A and B according to the observer inS? y:V= 0.6c -100 cmin- A' Flashbulb
-
Angina, Inc., has 5 million shares outstanding. The firm is considering issuing an additional 1 million shares. After selling these shares at their $20 per share offering price and netting 95% of the...
-
A system has two processes P1 and P2 with P1 having higher priority. They share an I/O device ADC. If P2 acquires the ADC from the RTOS and P1 becomes ready, how does the RTOS schedule the processes...
-
Sapporo K.K. was sued by a competitor in late 2017, and company management concluded that there was a 55 percent probability that the company would lose the lawsuit. The best estimate of the loss on...
-
Solve using MATLAB You should get same answers as worked out T=313 K 1) Use the Van der Waals EOS and calculate the PV value of CO2(g) (n-1 mol) at 313K: = b= 0.0427 mol P(atm) PV(obs.) (atm L/g)...
-
Suggest a mechanism for the oxo reaction (Eq. 18.63) involving intermediates that are consistent with the 16- and 18-electron rules. H,C=CH2 + H2 + CO ethylene HCO (CO)4 100-120 C CHCHCH...
-
Give the structure of the major product formed in each case when the reactant(s) shown undergo alkene metathesis in the presence of an appropriate ruthenium catalyst. (a) CHOH T HC=CHCHCHCHCHCH=CH x...
-
Explain the difference between tangible assets and intangible assets. Is it necessary to have different accounting rules for tangible and intangible assets?
-
Wello Contractors purchased construction equipment with an invoiced price of $800,000 from Capital Manufacturers on 1 January 20x1, and issued a two-year note to Capital Manufacturers. The interest...
-
On 1 January 20x1, Delphi Company issued 4% convertible bonds with a face value of $10,000,000 at par. The bond matured on 31 December 20x5. The bond was convertible into ordinary shares of Delphi...
-
On 1 January 20x1, ACO Corporation awarded fixed options to 100 employees to acquire 10,000 shares of the company under the following terms: (a) The exercise price was $5 per share (same as the...
-
On 1 April 20x6, ABC Company entered into a contract to sell 10,000 shares in XYZ Company for delivery on 30 June 20x6 at a price that is not below $3.00 per share and not higher than $4.00 per...
-
On 1 July 20x3, Alpha Company introduced a share option plan. Under the plan, one million share options with an estimated fair value of $4.3 million was granted to selected employees. The options...
-
A survey conducted by the Consumer Reports National Research Center reported, among other things, that women spend an average of 1.2 hours per week shopping online. Assume that hours per week...
-
What exactly is a prima facie duty? How does an ethic of prima facie duties differ from monistic and absolutist ethical theories?
-
Draw the two chair conformations of each compound and label the substituents as axial and equatorial. In each case, determine which conformation is more stable. (a) Cis-1-ethyl-2-isopropylcyclohexane...
-
Using what you know about the conformational energetics of substituted cyclohexanes, predict which of the two decalin isomers is more stable. Explain your reasoning.
-
Convert each Newman projection to the equivalent line-angle formula, and assign the IUPAC name. (a) (b) (c) (d) (e) (f) (g) (h) (i) (j) CH2CH3 CH2CH3 CH3 CH CH3 Br CH3 CH2CH3 CH2CHs Cl CH CH(CH32...
-
Each of the following is an independent Case involving the ownership of voting shares of CCPCs. All of the corporations have taxation years that end on December 31 and have only one class of shares....
-
Graphs of the cost C(x), revenue R(x) and the profit P(x), in thousands of dollars, are shown, where x is the number of thousands of items produced (a) Use the graph to find the formula for the...
-
Cold Duck Manufacturing Inc. buys most of its raw materials from a single supplier. This supplier sells to Cold Duck on terms of 3.5/20, net 60. Cold Duck's trade credit has a nominal annual cost of...

Study smarter with the SolutionInn App


