Give the structure of the major product formed in each case when the reactant(s) shown undergo alkene
Question:
Give the structure of the major product formed in each case when the reactant(s) shown undergo alkene metathesis in the presence of an appropriate ruthenium catalyst.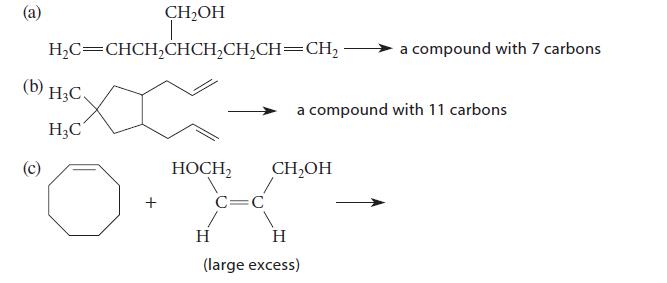
Transcribed Image Text:
(a) CH₂OH T H₂C=CHCH₂CHCH₂CH₂CH=CH₂ x (b) H3C. H₂C (c) + HOCH₂ CH₂OH C=C a compound with 11 carbons a compound with 7 carbons. H H (large excess)
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 100% (1 review)
a HOCH b H...View the full answer

Answered By

Bhartendu Goyal
Professional, Experienced, and Expert tutor who will provide speedy and to-the-point solutions. I have been teaching students for 5 years now in different subjects and it's truly been one of the most rewarding experiences of my life. I have also done one-to-one tutoring with 100+ students and help them achieve great subject knowledge. I have expertise in computer subjects like C++, C, Java, and Python programming and other computer Science related fields. Many of my student's parents message me that your lessons improved their children's grades and this is the best only thing you want as a tea...
3.00+
2+ Reviews
10+ Question Solved
Related Book For 

Question Posted:
Students also viewed these Sciences questions
-
Give the structure of the major alkene formed when the hydroxide of each of the following quaternary ammonium ions is heated. (a) (b) (CH3)3CCH2C(CH3)2 N(CH3)3 CH3 CH3CH2NCH2CH2CH2CH3 Kii CH
-
Give the structure of the major alkene formed when the hydroxide of each of the following quaternary ammonium ions is heated.
-
Give the structure of the cycloaddition product formed when benzyne is generated in the presence of furan. (See Section 11.21, if necessary, to remind yourself of the structure of furan.)
-
Consider a stylized two-period model with banking. The aggregate abatement cost function in period t is given by C(E)= (a t be) 2 /2b with a 1 < a 2 is D(E)=dE 2 /2. (a) Determine the optimal...
-
Two events in S are separated by a distance D = x 2 - x 1 and a time T = t 2 - t 1 . (a) Use the Lorentz transformation to show that in frame S, which is moving with speed V relative to S, the time...
-
Stacker Weight Loss currently pays an annual year-end dividend of $1.20 per share. It plans to increase this dividend by 5% next year and maintain it at the new level for the foreseeable future. If...
-
If you wanted to reduce the cache conflicts between the most computationally intensive parts of two processes, what are two ways that you could control the locations of the processes cache footprints?
-
A pharmaceutical company manufactures two drugs at Los Angeles and Indianapolis. The cost of manufacturing a pound of each drug depends on the location, as indicated in the file S13_42.xlsx. The...
-
The following units of an item were available for sale during the year: Beginning inventory Sale First purchase Sale Second purchase Sale 22 units @ $49 15 units @ $69 37 units @ $51 24 units @ $71...
-
Suggest an alkene metathesis reaction that would yield each of the following compounds as a major product. (c) F HC H (-)-citronellol (oil of roses) H D OH CHOH (b) COH
-
Show that the equilibrium mixture produced by alkene metathesis of two completely different alkenes with the following general structures contains 10 different alkenes. R H C=C H R + R H C=C H R4
-
In Problem 12, what would the risk-free rate have to be for the two stocks to be correctly priced relative to each other? Data From Problem 12 Stock Y has a beta of 1.05 and an expected return of 13...
-
The following information pertains to Causeway Company for the year ended 31 December 20x1: (a) Causeway Companys net profit attributable to ordinary shareholders for the year 20x1 was $8,000,000....
-
What is the overall net cash inflow/(outflow) arising from the forward contract? (a) $300,000 (b) ($300,000) (c) ($12,800,000) (d) ($12,500,000 On 1 October 20x4, SingCo committed to purchase...
-
In recent years, many firms have experienced significant difficulties in running their operations. This has primarily been down to the stagnant global economic environment, but is also a result of...
-
Ignore the previous question. On 1 April 20x4, XYZ Company entered into a commitment to sell 10,000 units of securities in S Company at a fixed price for delivery on 30 June 20x4. If the call option...
-
If XYZ Company purchased the call option primarily to benefit from an expected rising market value of the call option, which of the following statements best describes the appropriate accounting...
-
A Department of Transportation survey showed that 60% of U.S. residents over 65 years of age oppose use of cell phones in flight even if there were no issues with the phones interfering with aircraft...
-
Read the case study Richter: Information Technology at Hungarys Largest Pharma and answer the following question: How does the organization ensure the accuracy of the data it stores?
-
Draw Newman projections along the C3-C4 bond to show the most stable and least stable conformations of 3-ethyl- 2, 4, 4-trimethylheptane.
-
Conformational studies on ethane-1,2-diol (COCH2-CH2OH) have shown the most stable conformation about the central C-C bond to be the gauche conformation, which is 9.6 kJ/mol (2.3 kcal/mol) more...
-
The most stable form of the common sugar glucose contains a six-membered ring in the chair conformation with all the substituents equatorial. Draw this most stable conformation of glucose. HO glucose
-
Donna Corporation (the Company) purchased a piece of equipment on January 1, 2022, for a cash purchase price of $100,000. The equipment qualified for and the Company elected to depreciate the...
-
How does law in the United States integrate the four foundational legal elements? Constitutional Law Statutory Law Common Law Equity Law Select one type of law and explain how it is applied in the...
-
X-3 Identify restrictions on the domain of f(x)= X+2 X-3

Study smarter with the SolutionInn App


