Tert-butyl esters can be prepared by the acid-catalyzed reaction of methylpropene (isobutylene) with carboxylic acids. Suggest a
Question:
Tert-butyl esters can be prepared by the acid-catalyzed reaction of methylpropene (isobutylene) with carboxylic acids.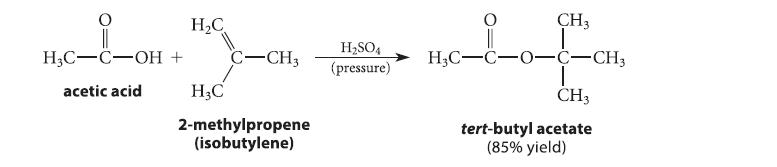
Suggest a mechanism for this reaction that accounts for the role of the acid catalyst.
Transcribed Image Text:
H3C-C-OH acetic acid H₂C + C-CH3 H3C 2-methylpropene (isobutylene) H₂SO4 (pressure) CH3 Lof H₂C-C-0-C-CH3 CH3 tert-butyl acetate (85% yield)
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 50% (2 reviews)
In this reaction the carboxylic acid is alkylated b...View the full answer

Answered By

Utsab mitra
I have the expertise to deliver these subjects to college and higher-level students. The services would involve only solving assignments, homework help, and others.
I have experience in delivering these subjects for the last 6 years on a freelancing basis in different companies around the globe. I am CMA certified and CGMA UK. I have professional experience of 18 years in the industry involved in the manufacturing company and IT implementation experience of over 12 years.
I have delivered this help to students effortlessly, which is essential to give the students a good grade in their studies.
3.50+
2+ Reviews
10+ Question Solved
Related Book For 

Question Posted:
Students also viewed these Sciences questions
-
The catalyst [Rh(Ph 2 PCH 2 CH 2 PPh 2 )] + can be prepared by the reaction of [Rh(nbd)(Ph 2 PCH 2 CH 2 PPh 2 )] + (nbd = 25.37) with two equivalents of H 2 . In coordinating solvents [Rh(Ph 2 PCH 2...
-
The hydrolysis of the ester shown here is catalyzed by morpholine, a secondary amine. Propose a mechanism for this reaction. (The pKa of the conjugate acid of morpholine is 9.3, so morpholine is too...
-
(a) Organolithium reagents such as methyllithium (CH 3 Li) react with carboxylic acids to give ketones. Two equivalents of the lithium reagent are required, and the ketone does not react further....
-
Thomas Gilbert and Susan Bradley formed a professional corporation called Financial Services Inc.A Professional Corporation, each taking 50 percent of the authorized common stock. Gilbert is a CPA...
-
The unadjusted trial balance and adjustment data of Smith Real Estate Appraisal Company at June 30, 2012, follow: Adjustment data at June 30, 2012: a. Prepaid insurance expired, $400. b. Accrued...
-
Money magazine (January 2007) reported that an average of 77 million adults in the United States make financial resolutions at the beginning of a new year. Consider the following frequency...
-
It would be helpful to me if I could start by asking you to basically tell me about your job. Okay?
-
Condensed financial data of Pat Metheny Company for 2008 and 2007 are presented below. Additional information: During the year, $70 of common stock was issued in exchange for plant assets. No plant...
-
New parents bring their 3-month-old to the clinic for a rash on the infants head. On examination, the skin affected by the rash is thickened, yellowish white in color, scaly, and looks waxy. In...
-
Give the structure of the ester formed when (a) Isobutyric acid reacts with diazomethane in ether. (b) Succinic acid reacts with a large excess of diazomethane in ether. (c) Isobutyric acid reacts...
-
(a) You learned in Sec. 19.7 that carbonyl-addition reactions can occur under basic conditions. The hydrolysis of methyl benzoate is also promoted by OH. Write a mechanism for the hydrolysis of...
-
Assume that the per-worker production function is y t = 2k t 0.5 . The saving and depreciation rates are estimated at 0.2 and 0.04, respectively. a) Calculate the capital-labor ratio steady state for...
-
Ying Import has several bond issues outstanding, each making semiannual interest payments. The bonds are listed in the following table. Bond Coupon Rate Price Quote Maturity Face Value 1 5.8 % 105.56...
-
2. What is the future value of an annuity retirement account if you deposit $2500 every year for 35 years? Assume the interest rate is 5.5% per year.
-
In 2013, Senate Democrats then in the majority triggered what was called the "nuclear option" for the first time. Frustrated with what they considered the relentless Republican obstruction of...
-
Establish the factors that must be taken into consideration and take into account the factors of sustainability, differentiation and growth?
-
A mass of 200 gms hangs from the rim of a wheel of radius 5 cm. Mass falls from rest to 2m in 5 sec. find the moment of inertia of the wheel?
-
Otsego Glass Company manufactures window glass for automobiles. The following data pertain to the Plate Glass Department. Work n process, June 1: Direct material...
-
Teasdale Inc. manufactures and sells commercial and residential security equipment. The comparative unclassified balance sheets for December 31, 2015 and 2014 are provided below. Selected missing...
-
The following chlorides (Ph = phenyl) undergo solvolysis in ethanol at the relative rates given in parentheses. How can you explain these results? Ph Ph Ph (3 x 106) (0.08) (300)
-
Provide a detailed mechanism for each of the following reactions. Include contributing resonance structures and the resonance hybrid for the arenium ion intermediates. (a) (b) (c) HNO HSO NO2 Br Bra,...
-
Provide a detailed mechanism for the following reaction. H,SO + H2O
-
For each critical audit matter identified in the current year's audit, PCAOB auditing standards require the auditor's report to include communication of all of the following, except for...
-
Frequency responses of a quarter car. A car is moving on a wavy road with a wave length d = 20 m and wave amplitude d = 0.08 m. Ms = 200 kg my = 40 kg kn = 220000 N/m ks = 8000 N/m C = 1000 Ns/m...
-
Once the trustee has determined the assets of a bankrupt, s/he can sell the remaining assets and then use the proceeds to pay out the remaining funds. The first parties paid from these funds are the...

Study smarter with the SolutionInn App


