The banned insecticide chlordane is reported to lose some of its chlorine and to be converted into
Question:
The banned insecticide chlordane is reported to lose some of its chlorine and to be converted into other compounds when exposed to alkaline conditions. Explain.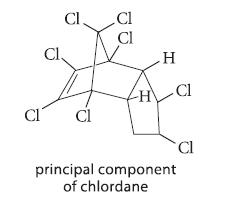
Transcribed Image Text:
CI CI Cl, Cl Cl CI H H\ Cl principal component of chlordane Cl
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 100% (QA)
In base some of the chlorines are eliminate...View the full answer

Answered By

Muhammad adeel
I am a professional Process/Mechanical engineer having a vast 7 years experience in process industry as well as in academic studies as a instructor. Also equipped with Nebosh IGC and lead auditor (certified).
Having worked at top notch engineering firms, i possess abilities such as designing process equipment, maintaining data sheets, working on projects, technical biddings, designing PFD and PID's etc.
Having worked as an instructor in different engineering institutes and have been involved in different engineering resrearch projects such as refinery equipment designing, thermodynamics, fluid dynamics, chemistry, rotary equipment etc
I can assure a good job within your budget and time deadline
4.90+
52+ Reviews
60+ Question Solved
Related Book For 

Question Posted:
Students also viewed these Sciences questions
-
A sample containing an alkali sulfate is dried, weighed and dissolved in dilute HCl. Barium chloride solution is added in excess to precipitate barium sulfate, and the precipitate is digested in the...
-
The Swift Company is planning to finance an expansion. The principal executives of the company agree that an industrial company such as theirs should finance growth by issuing common stock rather...
-
Chlordane is a powerful insecticide that was used in the United States during the second half of the twentieth century. Its use was discontinued in1988 in recognition of its persistence and...
-
A group of 70 students were asked to record the last digit of their social security number. a. Draw a relative frequency histogram using the values 0 through 9 as the class midpoints. What is the...
-
The state government established a capital project fund in 2011 to build new highways. The fund is supported by a 5 percent tax on diesel fuel sales in the state. The tax is collected by private gas...
-
Wilmington Chemical Company produces three products: ethylene, butane, and ester. Each of these products has high demand in the market, and Wilmington Chemical is able to sell as much as it can...
-
True or False: If \(E R R>M A R R\), then MIRR \(>\) MARR.
-
The comparative balance sheet of Merrick Equipment Co. for December 31, 2014 and 2013 is as follows: Additional data obtained from an examination of the accounts in the ledger for 2014 are as...
-
The adjusted trial balance for Chiara Company as of December 31 follows. Cash Accounts receivable Interest receivable Debit $ 30,000 Credit 52,000 18,000 nt ok Notes receivable (due in 90 days)...
-
When 2,3-dimethylbutane is treated with Br 2 in the presence of light, the bromine-containing compounds obtained in greatest amount are compound A (C 6 H 13 Br) and compound B (C 6 H 12 Br 2 )....
-
Propose a synthesis of ethyl neopentyl ether, C 2 H 5 OCH 2 C(CH 3 ) 3 , from an alkyl bromide and any other reagents.
-
On December 31, 2025, Oakbrook Inc. rendered services to Beghun Corporation at an agreed price of $102,049, accepting $40,000 down and agreeing to accept the balance in four equal installments of...
-
How infuential was early industrial psychology in increasing our knowledge of the behaviour of people in organizations?
-
What is the diference between behaviourism and psychoanalysis?
-
Discuss the difference between reinforcement and socialization in the context of a behavioural perspective on personality.
-
Discuss the difference between personality and intelligence.
-
Define the following terms: (a) compliant strategy; (b) internalexternal locus of control; (c) selfefcacy; (d) self-monitoring; (e) sensation-seeking; and (f) field dependencyindependency.
-
You and several of your classmates have just graduated from college and are evaluating various investment opportunities, including a start-up company that would produce high-quality jackets...
-
State whether each of the following will increase or decrease the power of a one-way between-subjects ANOVA. (a) The effect size increases. (b) Mean square error decreases. (c) Mean square between...
-
On catalytic hydrogenation over a platinum catalyst, ribose (Problem 9.57) is converted into ribitol. Is ribitol optically active or inactive?Explain. CH2 Ribitol
-
Hydroxylation of cis-2-hutene with OsO4 yields 2, 3-butanediol. What stereochemistry do you expect for the product?
-
Hydroxylation of trans-2-butene with OsO4 also yields 2, 3-butanediol. What stereochemistry do you expect for the product?
-
Enterprise Risk Management has been conceptualized in two dimensions, i.e. the types of risk and the various risk management process steps. Shoprite, one of the largest retailers in South Africa has...
-
PMI describes stewardship as being "a diligent, respectful, and caring steward steward". Stewards act responsibly to carry out activities with integrity, care, and trustworthiness while maintaining...
-
Image caption Sibling Company Issued $670,000 par value, 10-year bonds at 105 on January 1, 20X3, which Mega Corporation purchased. The coupon rate on the bonds is 9 percent. Interest payments are...

Study smarter with the SolutionInn App


