The following compound is unknown, but you are contemplating its synthesis and characterization. Predict its NMR spectrum
Question:
The following compound is unknown, but you are contemplating its synthesis and characterization. Predict its NMR spectrum under each of the following assumptions: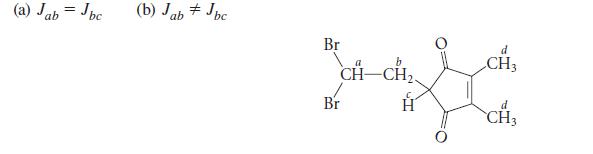
Transcribed Image Text:
(a) Jab = Jbc (b) Jab Jbc. Br b CH-CH₂ d CH3 Lich d CH3 Br
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 50% (2 reviews)
a b The resonance for protons He will be a singlet The resonances for protons H Hand He will al...View the full answer

Answered By

Asim farooq
I have done MS finance and expertise in the field of Accounting, finance, cost accounting, security analysis and portfolio management and management, MS office is at my fingertips, I want my client to take advantage of my practical knowledge. I have been mentoring my client on a freelancer website from last two years, Currently I am working in Telecom company as a financial analyst and before that working as an accountant with Pepsi for one year. I also join a nonprofit organization as a finance assistant to my job duties are making payment to client after tax calculation, I have started my professional career from teaching I was teaching to a master's level student for two years in the evening.
My Expert Service
Financial accounting, Financial management, Cost accounting, Human resource management, Business communication and report writing. Financial accounting : • Journal entries • Financial statements including balance sheet, Profit & Loss account, Cash flow statement • Adjustment entries • Ratio analysis • Accounting concepts • Single entry accounting • Double entry accounting • Bills of exchange • Bank reconciliation statements Cost accounting : • Budgeting • Job order costing • Process costing • Cost of goods sold Financial management : • Capital budgeting • Net Present Value (NPV) • Internal Rate of Return (IRR) • Payback period • Discounted cash flows • Financial analysis • Capital assets pricing model • Simple interest, Compound interest & annuities
4.40+
65+ Reviews
86+ Question Solved
Related Book For 

Question Posted:
Students also viewed these Sciences questions
-
The following compound is known to be chiral. Draw its enantiomer, and explain the source of chirality. CH
-
The mass spectrum of unknown compound A shows a molecular ion at m/z 116 and prominent peaks at m/z 87 and m/z 101. Its UV spectrum shows no maximum above 200 nm. The IR and NMR spectra of A follow....
-
Compound A has molecular formula C 8 H 8 O. An IR spectrum of compound A exhibits a signal at 1680 cm -1 . The 1 H NMR spectrum of compound A exhibits a group of signals between 7.5 and 8 ppm (with a...
-
A $250 suit is on sale for $190, and a $90 pair of shoes is on sale for $65. Find the average percent decrease in price for the 2 items.
-
Relating market value to book value of shareholders' equity Firms prepare their balance sheets using authoritative guidance for the recognition and measurement of assets and liabilities. Accountants...
-
During 2010, Howard Company purchased land for $375,000. It paid $125,000 in cash and signed a $250,000 mortgage for the rest. The company also sold for $95,000 cash a building that originally cost...
-
All blocks in Figure P8.47 are identical and you can ignore any friction in the pulleys. Rank the configurations in increasing order of tension in the rope. (Use free-body diagrams.) Data from Figure...
-
Dakota Manufacturing Inc. normally produces 10,000 units of product A each month. Each unit requires 4 hours of direct labor, and factory overhead is applied on a direct labor hour basis. Fixed costs...
-
1. Can you give examples of any successful OD programmes? Draw on recent research for your answer. 2. You have been asked to give a talk to a postgraduate management conference on The qualities...
-
When 3-bromopropene is allowed to react with HBr in the presence of peroxides, a compound A is formed that has the following NMR spectrum: 3.60 (4H, t, J = 6 Hz); 2.38 (2H, quintet, J = 6 Hz). (a)...
-
Give the structure of a compound C 7 H 16 O 3 with the following NMR spectrum: 1.30 (3H, s); 1.93 (2H, t, J = 7.3 Hz); 3.18 (6H, s); 3.33 (3H, s); 3.43 (2H, t, J = 7.3 Hz). Its IR spectrum shows...
-
Use the definition of the derivative and the trigonometric identity cos (x + h) = cos x cos h - sin x sin h to prove that d/dx (cos x) = -sin x.
-
Why would an organization have a large number of days cash on hand? How could its strategic plans influence this number?
-
What role does a primary key play in performing a data join?
-
How does product differentiation affect the level of rivalry in a market?
-
Why is it important to consistently evaluate data that reflect how well key stakeholders needs are being met?
-
What makes change so difficult for many organizations?
-
A study was made to compare the costs of supporting a family of four Americans for a year in different foreign cities. The lifestyle of living in the United States on an annual income of $75,000 was...
-
Design an experiment to demonstrate that RNA transcripts are synthesized in the nucleus of eukaryotes and are subsequently transported to the cytoplasm.
-
What is dn for each of the following complexes? Pd(PPh3)4
-
Within set, rank the compounds in order of increasing rates of their SN2 reactions. Explain your reasoning. 1-bromocyclohexene, bromocyclohexane, 1-(bromomethyl)cyclohexene
-
How many CO ligands would be accommodated by Fe(O) if we assume that the resulting complex follows the 18-electron rule?
-
S Inc installs complex satellite navigation systems in cars, at avery large national depot. The standard cost of an installation is shownbelow. The budgeted volume is 1,000 units installed each...
-
4. (15 points) The final simplex table for the LP relaxation of an IP is given below, where s and 82 are the slack variables corresponding to the first and second con- straints of the original model....
-
Teachers can't understand why Student A is doing very poorly in class. They referred the student to a psychologist so that an intelligence or aptitude assessment could be conducted. Surprisingly the...

Study smarter with the SolutionInn App


