The following molecule has a barrel shape (in which the benzene rings are the walls of the
Question:
The following molecule has a barrel shape (in which the benzene rings are the “walls” of the barrel). It forms a noncovalent complex with the iodide salt of acetylcholine in chloroform solvent.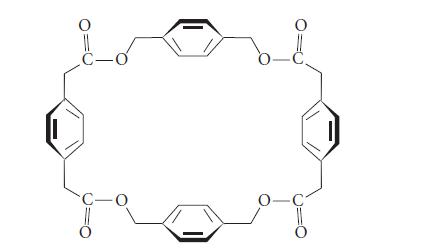
Describe the orientation of the acetylcholine molecule within the complex.
Transcribed Image Text:
O C-o C-O 8 O -C₂ 0-C
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 100% (1 review)
The positive end of the acetylcholine ion would orient its...View the full answer

Answered By

Madhur Jain
I have 6 years of rich teaching experience in subjects like Mathematics, Accounting, and Entrance Exams preparation. With my experience, I am able to quickly adapt to the student's level of understanding and make the best use of his time.
I focus on teaching concepts along with the applications and what separates me is the connection I create with my students. I am well qualified for working on complex problems and reaching out to the solutions in minimal time. I was also awarded 'The Best Tutor Award' for 2 consecutive years in my previous job.
Hoping to get to work on some really interesting problems here.
5.00+
1+ Reviews
10+ Question Solved
Related Book For 

Question Posted:
Students also viewed these Sciences questions
-
The N-methylquinolinium ion forms a noncovalent complex with molecule A in water that has a standard free energy of dissociation G d 5 28.9 kJ mol 1 (6.9 kcal mol 1 ). The neutral molecule...
-
Identify the process evaluation article that you chose and explain why you selected this example. Describe the purpose of the evaluation, the informants, the questions asked, and the results of the...
-
Which choice is greener in a chemical process? Explain. (a) Benzene as a solvent or water as a solvent. (b) The reaction temperature is 500 K, or 1000 K. (c) Sodium chloride as a by-product or...
-
What are some Marketing Strategies for Delivering Objectives Polestar (Car company) has used or using?
-
What is the maximum penalty and prison term that can be charged to a CEO and/or CFO under the Sarbanes-Oxley Act?
-
Benson Enterprises is evaluating alternative uses for a three-story manufacturing and warehousing building that it has purchased for $1,450,000. The company can continue to rent the building to the...
-
Diageo North America, Inc., the owner of the Bulleit brand family of whiskeys, sued competitor W.J. Deutsch & Sons Ltd. for allegedly infringing upon the trade dress of Diageos Bulleit bottles....
-
Refer to the Hungry Dawg Restaurant example presented in this chapter. Health claim costs actually tend to be seasonal, with higher levels of claims occurring during the summer months (when kids are...
-
There are four types of relational models: communal sharing, authority ranking, equality matching, and market pricing. Which relational models do you use in your interpersonal interactions? Is one...
-
Using the Hckel 4n + 2 rule, determine whether each of the following compounds is likely to be aromatic. Explain how you arrived at the -electron count in each case. (a) (c) CO (b) -C=C- (d) :1
-
Which of the compounds or ions in Problem 15.38 are likely to be antiaromatic? Explain. Problem 15.38 Which of the following species should be aromatic by the Hckel 4n + 2 rule? (a) thiophene G O H H...
-
A block diagram for a divider that divides an 8-bit unsigned number by a 4-bit unsigned number to give a 4-bit quotient is shown subsequently. The Xi inputs to the subtractors are shifted over one...
-
The reason many homeowners cannot sell their homes when housing prices are falling is because ______. a) there are no buyers in the market b) there are too many houses on the market c) no one can get...
-
When market price is above equilibrium price, _______. a) market price will rise b) equilibrium price will rise c) market price will fall d) equilibrium price will fall
-
When supply rises and demand stays the same, _______. a) equilibrium quantity rises b) equilibrium quantity falls c) equilibrium quantity stays the same
-
At equilibrium price, quantity demanded is _____. a) greater than quantity supplied b) equal to quantity supplied c) smaller than quantity supplied
-
At equilibrium, quantity demanded is __________equal to quantity supplied. a) sometimes b) always c) never
-
Jonas owns a building that he leases to Dipper, Inc., for $5,000 per month. The owner of Dipper has been complaining about the condition of the restrooms and has proposed making improvements that...
-
Write the general quadratic equation y2 - 8y - 4x + 28 = 0 in standard form. Determine the vertex, focus, and directrix of the parabola defined by this equation. Sketch a graph.
-
Propose structures for compounds that meet the following descriptions: (a) C5H8, with IR absorptions at 3300 and 2150 cm1 (b) C4H80, with a strong IR absorption at 3400 cm1 (c) C4H80, with a strong...
-
How could you use infrared spectroscopy to distinguish between the following pairs of isomers? (a) HC CCH2NH2 and CH3CH2C N (b) CH3COCH3 and CH3CH2CHO
-
Two infrared spectra are shown. One is the spectrum of cyclohexane, and the other is the spectrum of cyclohexane. Identify them, and explain youranswer. (a) 100 80 60 20 - 1000 4000 3500 3000 2500...
-
1. Uber is a business dealing the rental of vehicles. It has over 50 million users in the USA alone. In 2016 hackers stole data of the over 50 million users. A demand notice was issued and Uber had...
-
three months ago CruiseEAstern Pty Ltd, a cruise shipping business, borrowed $1 billion from the Federation Bank on security over CruiseEastern Pty Ltd's fleet of cruise ships. CruiseEastern was...
-
PROBLEM 827 Completing a Master Budget [LO82, LO84, LO87, LO88, LO89, LO810] The following data relate to the operations of Shilow Company, a wholesale distributor of consumer goods: Current assets...

Study smarter with the SolutionInn App


