The G difference between the axial and equatorial conformations of methylcyclohexane (7.4 kJ mol 1 , 1.74
Question:
The ΔG° difference between the axial and equatorial conformations of methylcyclohexane (7.4 kJ mol–1, 1.74 kcal mol–1; see Fig. 7.9) is about the same as the ΔH° difference. Calculate the percentages of axial and equatorial conformations present in one mole of methylcyclohexane at 25 °C.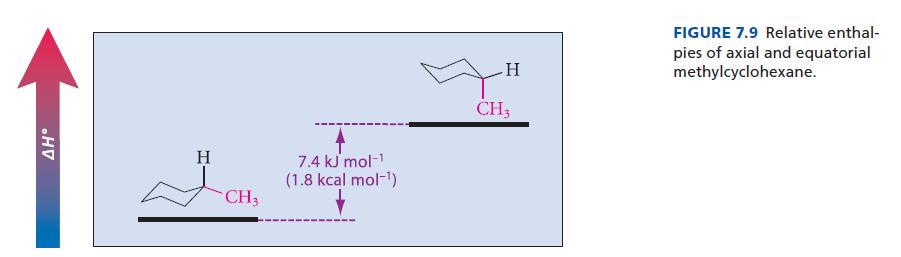
Transcribed Image Text:
AH° H CH3 7.4 kJ mol-¹ (1.8 kcal mol-¹) H CH3 FIGURE 7.9 Relative enthal- pies of axial and equatorial methylcyclohexane.
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 100% (5 reviews)
We apply text Eq 336b text p 110 with G 74 kJ mol 1 Letting the axial fo...View the full answer

Answered By

PALASH JHANWAR
I am a Chartered Accountant with AIR 45 in CA - IPCC. I am a Merit Holder ( B.Com ). The following is my educational details.
PLEASE ACCESS MY RESUME FROM THE FOLLOWING LINK: https://drive.google.com/file/d/1hYR1uch-ff6MRC_cDB07K6VqY9kQ3SFL/view?usp=sharing
3.80+
3+ Reviews
10+ Question Solved
Related Book For 

Question Posted:
Students also viewed these Sciences questions
-
A loan of $15,000 is compounded monthly at 4.5% and is to be paid off with monthly payments for 3 years. find the monthly payment PMT.
-
Table 3-6 shows that the axial-equatorial energy difference for methyl, ethyl, and isopropyl groups increases gradually: 7.6, 7.9, and 8.8 kJ mol (1.8, 1.9, and 2.1 kcal mol). The tertbutyl group...
-
The following additional information is available for the Dr. Ivan and Irene Incisor family from Chapters 1-5. Ivan's grandfather died and left a portfolio of municipal bonds. In 2012, they pay Ivan...
-
Assume an employee signs a non - disparagement clause. What would be considered breaking this clause? a . ) Telling a company's trade secret to a competing firm for a fee b . ) Using a work computer...
-
What are the steps for a court in statutory interpretation?
-
Jacqui and Joanne plan to buy a bed-and-breakfast inn for $200,000. Jacqui will contribute $20,000 toward the purchase and operate the enterprise. Joannes primary role is that of investor. She will...
-
A construction contract has the following language: It is the responsibility of the contractor to inspect and become familiar with the Project and to acquaint itself thoroughly with all conditions...
-
Presented below are two independent situations. 1. Chicory Cosmetics acquired 15% of the 200,000 shares of common stock of Racine Fashion at a total cost of $13 per share on March 18, 2012. On June...
-
You are the Director of the 6 ICU units at your facility. You have an all RN staff and CNA's in each of the units. Your CNO has recently informed you that he has performed a supply audit and has...
-
(a) The axial conformation of fluorocyclohexane is 1.0 kJ mol 1 (0.25 kcal mol 1 ) less stable than the equatorial conformation. What is the energy cost of a 1,3-diaxial interaction between hydrogen...
-
Given that the twist-boat form is 15.9 kJ mol 1 (3.8 kcal mol 1 ) higher in standard free energy than the chair form of cyclohexane, calculate the percentages of each form present in a sample of...
-
Consider a two-phase mixture of ammonia and water in equilibrium. Can this mixture exist in two phases at the same temperature but at a different pressure?
-
List what the Sonic Boom company is currently doing to get shoppers to advocate for them digitally List at least two recommendations for the Sonic boom to get shoppers to advocate for them digitally...
-
The following UN Sustainability Goals are covered GOAL 4: Quality Education GOAL 5: Gender Equality GOAL 8: Decent Work and Economic Growth GOAL 9: Industry, Innovation and Infrastructure GOAL 10:...
-
To evaluate extra revenues collected from additional costs _____ analysis is used Select one: a. Qualitative b. Quantitative O c. Marginal analysis d. Cost effectiveness analysis
-
A nurse and long-time advocate for quality nursing care for the elderly is planning to open a small nursing facility in town to better meet the needs of the community with financial support from many...
-
Imagine that your company is considering the launch of a product or brand in a new market. What company and product are you choosing? SWOT analysis for the company. Strengths In light of the...
-
In long-run equilibrium, P = minimum ATC = MC. Of what significance for economic efficiency is the equality of P and minimum ATC? The equality of P and MC? Distinguish between productive efficiency...
-
C- Consider the following scenario:- A supermarket needs to develop the following software to encourage regular customers. For this, the customer needs to supply his/her residence address, telephone...
-
Rank the following substances in order of increasing acidity: C CH3CH3 CHCCH2CCH Acetic acid (pKa = 4.76) Phenol Acetone (pKa = 19.3) 2,4-Pentanedione (pKa = 9) (pKa = 9.9) %3D %3D
-
Which, if any, of the four substances in Problem 2.43 is a strong enough acid to react almost completely with NaOH? (The pKa of H2O is 15.74.)
-
The ammonium ion (NH4+, pKa = 9.25) has a lower pKa then the methyl-ammonium ion (CH3NH3+, pKa = 10.66). Which is the stronger base, ammonia (NH3) or methylamine (CH3NH2)? Explain
-
Beyonce went to the mall and saw a massage chair that she would have to take a loan out for $6,500 to purchase. The bank said that she could get a simple interest rate of 8% for 5 years. What is the...
-
Solve the equation 5.3g+9=2.39 + 15. a. Find the value of g. b. Explain how you can check that the value you found for g is correct. If your check does not work, does that mean that your result is...
-
1. (20%) Let S 2+5+8+11 +...+998 a) Express S using the summation notation (). b) Determine the explicit formula for S and then evaluate the sum. Sow your work to receive full credit.

Study smarter with the SolutionInn App


