Rank the following substances in order of increasing acidity: C CH3CH3 CHCCH2CCH Acetic acid (pKa = 4.76)
Question:
Rank the following substances in order of increasing acidity:
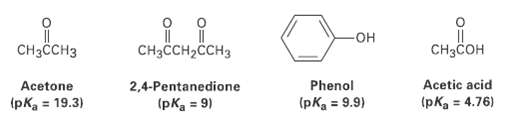
Transcribed Image Text:
Cнзсон CH3CH3 CHзCCH2CCHз Acetic acid (pKa = 4.76) Phenol Acetone (pKa = 19.3) 2,4-Pentanedione (pKa = 9) (pKa = 9.9) %3D %3D
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 76% (17 reviews)
The substances with ...View the full answer

Answered By

Jayshree Rathi
Hello Students!
This is Jayshree Rathi. I work on a number of renowned student-centric channels such as Chegg, coursehero, as a certified private tutor.
If you are looking for relevant and original content to complete your assignments, essays, and homework, then contact me and within the promised time, I will deliver you your personalized academic work and help you score the best.
4.80+
1+ Reviews
10+ Question Solved
Related Book For 

Question Posted:
Students also viewed these Organic Chemistry questions
-
Rank the following substances in order of increasing acidity: (a) (CH3)2CHOH, HC CH, (CF3)2CHOH, CH4OH (b) Phenol, p-methyl phenol, p-(trifluoromethyl) phenol (c) Benzyl alcohol, phenol, p-hydroxy...
-
Rank the following in order of increasing acidity for the indicated hydrogen atoms (bold) (1 = least acidic; 4 = most acidic). (a) (b) (c) O C OCH3 . CH3 H3C
-
Rank the following substances in order of their expected SN1 reactivity: Br Br H CHCCH CHCH2Br H2C=CHBr
-
Prepare adjusting journal entries, as needed, considering the account balances excerpted from the unadjusted trial balance and the adjustment data. A. depreciation on buildings and equipment, $17,500...
-
Individuals who receive substantial Social Security benefits are usually not eligible for the tax credit for the elderly or disabled because these benefits effectively eliminate the base upon which...
-
You are designing a two-string instrument with metal strings 35.0 cm long, as shown in Fig. P15.58. Both strings are under the same tension. String S 1 has a mass of 8.00 g and produces the note...
-
There is a stereotype that men are willing to talk about something, even if they admit that they dont know anything about that something. Using the PewSocial-Media dataset, first select cases so that...
-
1. In relation to Section 15.3, what were the types of change pursued at Fiat and Chrysler? Were these appropriate to the change contexts? 2. How would you describe the leadership style of Sergio...
-
P5-9 (similar to) Question Help (Related to Checkpoint 5.2) (Future value) You have just introduced "must have" headphones for the iPod. Sales of the new product are expected to be 30,000 units this...
-
1. Of the 10 psychological phenomena introduced in Chapter 1, identify which ones apply to the minicase, and give reasons to support your answer. 2. The Times article notes that three days after the...
-
Maleic acid has a dipole moment, but the closely related fumaric acid, a substance involved in the citric acid cycle by which food molecules are metabolized, does not.Explain - C-OH - C=C c=C ...
-
Which, if any, of the four substances in Problem 2.43 is a strong enough acid to react almost completely with NaOH? (The pKa of H2O is 15.74.)
-
In response to the deregulation of the European airline industry during the 1980s and 1990s, European airlines followed their US peers in starting frequent flyer programs as a way to differentiate...
-
Nelsie Corporation has an outstanding 60-day 6% note receivable amounting to P 15,000 dated December of the ne year. The company is using the calendar year in preparing its financial statements. What...
-
Which resource is the bottleneck? What is the overall capacity of the orthopedist's office in patients/hour?
-
What are the comprehensive strategic implentation issues of Kmart with reference
-
1. Create both the written plan and the educational material to help African American women age 65+ control high blood pressure, take the special circumstances into consideration for the plan. 2. For...
-
Write down D & S equations for wireless phones; include two exogenous variables in each equation.
-
Which statement is least appropriate? A loss of direction can be the difference between a good audit and a boring report. Demotivated auditors are a problem for audit management even where they do...
-
Choose a company from the SEC EDGAR Web site for your Key Assignment to evaluate for the impact of convergence to IFRS. Review the financial reports and notes of the company you have chosen from the...
-
Redo Problem 4.45 if ethylene is described by the Peng-Robinson equation of state. Problem 4.45 The second virial coefficient B can be obtained from experimental PVT data or from an equation of state...
-
Assign the configurations of the compounds represented by these Fischer projections as R or S. CH -CH2CH3 b) CH2=CH- ) >N- CH-OH CH3 Serine
-
Explain whether each of these compounds is chiral or not: a) d) H C H c=c=c CHCH CCH HC PhwSi-OCH-CH b) CHCH Ph-N-CHCH, HC Br H C CH30 CH CO H c) f) H H HC CH30 CCH CH3 COH CH3 CO H
-
Although this biphenyl is chiral, it cannot be resolved. Explain. HO-C CH30 CO H OCH
-
Present Value Computations Using the present value tables, solve the following. ( Click here to access the PV and FV tables to use with this problem. ) Round your answers to two decimal places....
-
A company provided the following data: Sales $887,000 Variable costs $546,800 Fixed costs $310,000 Expected production and sales in units 36,000 What is the break-even point in sales dollars? Please...
-
How to solve them..equation and explain ..please.. 1. Selected information from the companys financial records is presented below Equipment, December 31, 2013 $300,000 Equipment, December 31, 2014...

Study smarter with the SolutionInn App


