The two most common forms of glucose, -D-glucopyranose and -D-glucopyranose, can be brought into equilibrium by dissolving
Question:
The two most common forms of glucose, α-D-glucopyranose and β-D-glucopyranose, can be brought into equilibrium by dissolving them in water with a trace of an acid or base catalyst.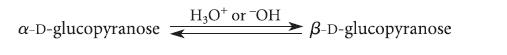
The specific rotation of the equilibrium mixture is 152.7 deg mL g–1 dm–1. The specific rotation of pure a-d-glucopyranose is 1112 degrees mL g–1 dm–1, and that of pure b-d-glucopyranose is 118.7 degrees mL g–1 dm–1.
What is the percentage of each form in the equilibrium mixture?
Transcribed Image Text:
a-D-glucopyranose H₂O* or "OH B-D-glucopyranose
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 0% (2 reviews)
Each compound contributes to the optical activity in prop...View the full answer

Answered By

Aysha Ali
my name is ayesha ali. i have done my matriculation in science topics with a+ . then i got admission in the field of computer science and technology in punjab college, lahore. i have passed my final examination of college with a+ also. after that, i got admission in the biggest university of pakistan which is university of the punjab. i am studying business and information technology in my university. i always stand first in my class. i am very brilliant client. my experts always appreciate my work. my projects are very popular in my university because i always complete my work with extreme devotion. i have a great knowledge about all major science topics. science topics always remain my favorite topics. i am also a home expert. i teach many clients at my home ranging from pre-school level to university level. my clients always show excellent result. i am expert in writing essays, reports, speeches, researches and all type of projects. i also have a vast knowledge about business, marketing, cost accounting and finance. i am also expert in making presentations on powerpoint and microsoft word. if you need any sort of help in any topic, please dont hesitate to consult with me. i will provide you the best work at a very reasonable price. i am quality oriented and i have 5 year experience in the following field.
matriculation in science topics; inter in computer science; bachelors in business and information technology
_embed src=http://www.clocklink.com/clocks/0018-orange.swf?timezone=usa_albany& width=200 height=200 wmode=transparent type=application/x-shockwave-flash_
4.40+
11+ Reviews
14+ Question Solved
Related Book For 

Question Posted:
Students also viewed these Sciences questions
-
The two most common marketing tools used for product advertising are ads on television and ads in a print magazine. Consumers' attitudes toward television and magazine advertising were investigated...
-
The January 2008 edition of Strategic Finance includes an article by Curtis C. Verschoor titled ERC Says Ethics Risk Landscape Still Treacherous. Instructions Read the article and answer the...
-
The two most common isotopes of uranium are 235U and 238U. (a) Compare the number of protons, the number of electrons, and the number of neutrons in atoms of these two isotopes. (b) Using the...
-
DTG agreed to merge with the SPQ. The total equity purchase price of $45,000 is going to be 50% financed issuing DTGs common shares and 50% financed with cash raised by DTG issuing long-term debt....
-
1. The Colorado statute prohibits assisting suicide. Did Dick Bauer violate the statute? 2. If you were on the jury, would you vote to convict?
-
Current reporting standards require the consolidated entity to include all the revenues, expenses, assets, and liabilities of the parent and its subsidiaries in the consolidated financial statements....
-
Barrett, Inc., restores antique automobiles. The retained earnings balance of the corporation was \($28,500\) at December 31,2009. During 2010, the corporation paid \($35,000\) in dividends to its...
-
Using the information in E22-3, assume that in July 2014, Thome Company incurs The following manufacturing overhead costs. Instructions (a) Prepare a flexible budget performance report, assuming that...
-
Exercise: Consider the equation x5 + 4x-7= 0. (a) Show that there is root between 1 and 2. (b) Estimate the root by the Bisection method.
-
(a) 2,3,4-Trichloropentane has two meso stereoisomers. Starting with the template below for each, complete line-and-wedge structures for the two meso stereoisomers. (b) Show the symmetry element in...
-
(S)-(1)-Aspartic acid is one of the naturally occurring a-amino acids. Over time in the environment or in aqueous solution, aspartic acid can undergo racemization very slowly (by a process that we...
-
The passenger-side side-view mirror on most cars is a convex mirror. (The warning that objects may be closer than they appear indicates that the mirror is convex.) a. View some object of known height...
-
As a Financial Planning student how will you explain to the Canadian public to increase their financial literacy with the topic Why should individuals and families have Financial Plan?
-
While analyzing Winter Company's 20Y1 and 20Y2 financial statements, you note the following: (000s omitted) Sales Cost of goods sold Gross profit SG&A expenses Accounts receivable Inventory Accounts...
-
Could you please explain the talefilm of canada in detail like it's key senior management positions, middle management positions, non management positions and support staff in details?
-
For Ulta Beauty Inc, Assess the company's operational budget -Based on the data, what is the overall current financial condition of the company
-
What type of organizational structure do you believe is most adaptable and responsive and discuss why. Consider the different organizational structures of functional structures, multi-divisional...
-
Refer again to the data from the MBA new-matriculants survey in exercise. a. Given that a person applied to more than one school, what is the probability that the person is 2426 years old? b. Given...
-
You are the newly appointed tax practitioner to complete Emilys tax return and have downloaded the prefill report for Emilys tax return (hint, you can read what a prefill report is here (Links to an...
-
Styrene, the simplest alkenylbenzene, is prepared commercially for use in plastics manufacture by catalytic dehydrogenation of ethylbenzene. How might you prepare styrene from benzene using reactions...
-
How would you prepare diphenylmethane, (Ph) 2Ch2, from benzene and an acid chloride?
-
Propose syntheses of the following substances from benzene: (a) m-Chloronitrobenzene (b) m-Chloroethylbenzene (c) 4-Chloro-1-nitro-2-propylbenzene (d) 3-Bromo-2-methylbenzenesulfonic acid
-
Image caption Develop a minimum 700-word branding strategy and marketing communication plan in Microsoft Word. This document should address at least 5 elements of the Situational Analysis and the...
-
Please identify one retail brand that (in your opinion) practices Relationship Marketing and one retail brand that practices Transactional Marketing. Support by citing TWO reasons why each are...
-
During this pandemic, How the brand like a football team think should improve its customer retention marketing. Recommend the steps that this brand needs to take to strengthen its digital CRM in...

Study smarter with the SolutionInn App


