Use the structure of the DielsAlder adduct to deduce the structure of the product X in the
Question:
Use the structure of the Diels–Alder adduct to deduce the structure of the product X in the reaction given in Fig. P15.82. Then give a curved-arrow mechanism for the formation of X.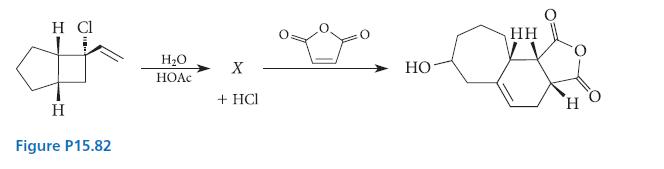
Transcribed Image Text:
H Cl да Н Figure P15.82 H2O НОАс X + HCI НО- HH
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 66% (3 reviews)
Follow the procedure used in solving Problem 1581 mentally rever...View the full answer

Answered By

Asd fgh
sadasmdna,smdna,smdna,msdn,masdn,masnd,masnd,m asd.as,dmas,dma.,sd as.dmas.,dma.,s ma.,sdm.,as mda.,smd.,asmd.,asmd.,asmd.,asm
5.00+
1+ Reviews
15+ Question Solved
Related Book For 

Question Posted:
Students also viewed these Sciences questions
-
In 1991, chemists at Rice University reported that they had trapped an unstable compound called spiropentadiene using its Diels-Alder reaction with excess 1, 3- cyclopentadiene, giving the product in...
-
2. Draw the curved arrow mechanism for the reaction between 2-methoxyphenoxide and 3-chloro-1,2-propanediol, in the guaifenesin synthesis. Be sure to clearly show the bimolecular transition state.
-
Draw the simplified curved arrow mechanism for the mechanistic step of (E) -3,5-dimethylhept-3-em-2-one and (CH 3 CH 2 ) 2 CuLi to give the major charged species which is formed. Draw all electrons...
-
Some managers and union officials also refer to the collective agreement as what? Multiple Choice The real boss The rule book The bible The guiding framework Strategic doctrine
-
Slocum Inc. has one product line that is unprofitable. What circumstances may cause overall company net income to be lower if the unprofitable product line is eliminated?
-
Discuss the advantages and disadvantages of having the audit committee deal directly with the external auditors.
-
Jerome M. Eisenberg is an antiquities dealer and a self-proclaimed expert in classical antiquities with a doctorate in Roman, Egyptian, and Near Eastern art. Maurice E. Hall Jr. is an art dealer who...
-
The Gorman Manufacturing Company must decide whether to manufacture a component part at its Milan, Michigan, plant or purchase the component part from a supplier. The resulting profit is dependent...
-
A 480-g iron pan is heated on a stove to a temperature of 130 C. The pan is then dropped into a partially-filled sink of water at 20 C. The final temperature reached by the sink and the pan is 40 C....
-
Account for the fact that the central benzene ring of [4]phenylene (Fig. P15.86) undergoes catalytic hydrogenation readily under conditions usually used for ordinary alkenes, but the other benzene...
-
Account for each of the transformations shown in Fig. P15.84 with a curved-arrow mechanism. (Dont try to explain any percentages.) In part (d), identify X; give the mechanisms for both the formation...
-
The unadjusted trial balance for Swift Creek Engineering at its year end, December 31, 2014, is as follows: Additional information: 1. Revenue of $1,440 was earned but unrecorded as at December 31,...
-
Why have various business groups found it necessary to establish codes of ethics?
-
How are the master budgets of a retail business and a service business similar to each other? How are they different from each other?
-
How would you describe the similarities and differences between management accounting and financial accounting? Why are they different, and why are they similar?
-
On the cash budget, why is the beginning cash balance for July the same as the beginning cash balance for the first quarter of the year? Why is the September ending cash balance the same as the first...
-
Given what you have learned from this chapter, how would you define accounting?
-
On January 1, Wilton loans Andy $90,000. The loan is to be repaid in 5 years with no interest charged. The applicable federal rate is 5%. Discuss the treatment of the loan for both Wilton and Andy in...
-
Gordon and Lisa estimate that they will need $1,875,000 in 40 years for their retirement years. If they can earn 8 percent annually on their funds, how much do they need to save annually?
-
Which compound in each of the following pairs is more basic? (a) CH 3 CH 2 NH 2 or CH 3 CH 2 CONH 2 (b) NaOH or CH 3 NH 2 (c) CH 3 NHCH 3 or pyridine
-
What product would you expect from Hofmann elimination of a heterocyclic amine such as piperidine? Write all thesteps? Piperidine
-
Classify each of the following monosaccharide?s: la) 0 (c) (b) (d) CH- . H- C=0 C=0 -- -- -- -- -- -- -- -- -- H2OH -- CH- Ribulose CH Threose CH- Tagatose 2-Deoxyribose
-
Mary Berry is evaluating two mutually exclusive projects (expected cash flows shown below). The firm's cost of capital is 11 percent. (Round each amount to the nearest cent.) Year Project A Project B...
-
Valuing a company using FCFF instead of FCFE is likely more accurate if reliable market value estimates of its debt and equity securities are available. Why or why not ? Explain
-
The Vancouver Fraser Port Authority is planning out their internships for the next calendar year. They would like to have 3 student interns working from Jan - Apr, 5 students working from May - Aug,...

Study smarter with the SolutionInn App


