Account for each of the transformations shown in Fig. P15.84 with a curved-arrow mechanism. (Dont try to
Question:
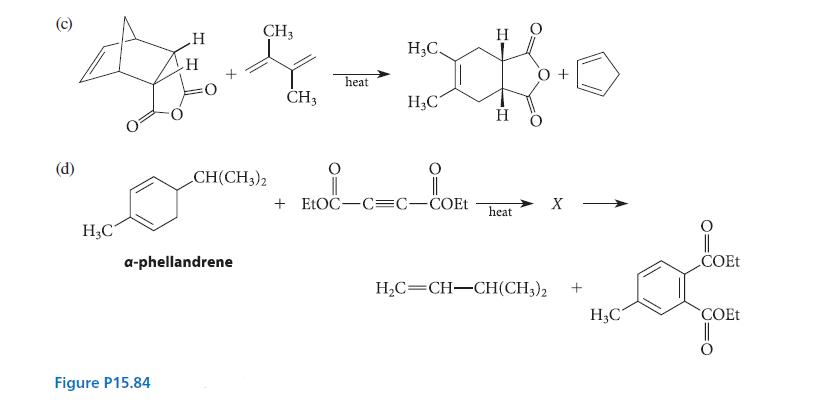
Account for each of the transformations shown in Fig. P15.84 with a curved-arrow mechanism. (Don’t try to explain any percentages.) In part (d), identify X; give the mechanisms for both the formation and the subsequent reaction of X; and explain why the equilibrium for the reaction of X strongly favors the products.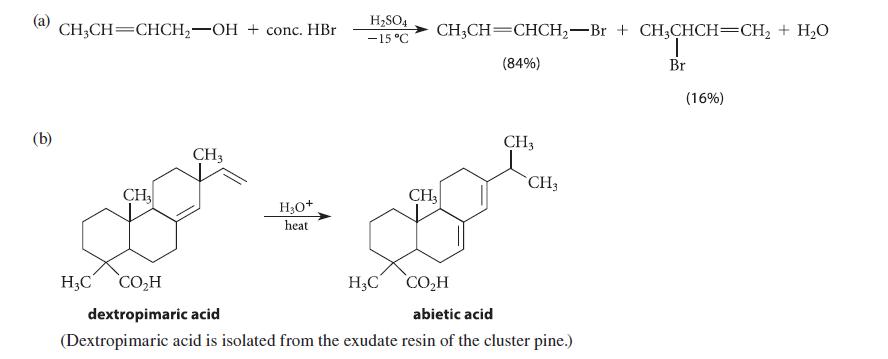
Transcribed Image Text:
(a) (b) CH3CH CHCH₂-OH conc. HBr CH3 H3C CO₂H CH3 H₂O+ heat H₂SO4 -15 °C CH₂CH=CHCH₂-Br + CH3CHCH=CH₂ + H₂O T (84%) Br H₂C CH3 CH3 CO₂H dextropimaric acid abietic acid (Dextropimaric acid is isolated from the exudate resin of the cluster pine.) CH3 (16%)
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 100% (1 review)
a b The mechanism below is shown beginning with the protonated alcohol which is formed under the str...View the full answer

Answered By

Somshukla Chakraborty
I have a teaching experience of more than 4 years by now in diverse subjects like History,Geography,Political Science,Sociology,Business Enterprise,Economics,Environmental Management etc.I teach students from classes 9-12 and undergraduate students.I boards I handle are IB,IGCSE, state boards,ICSE, CBSE.I am passionate about teaching.Full satisfaction of the students is my main goal.
I have completed my graduation and master's in history from Jadavpur University Kolkata,India in 2012 and I have completed my B.Ed from the same University in 2013. I have taught in a reputed school of Kolkata (subjects-History,Geography,Civics,Political Science) from 2014-2016.I worked as a guest lecturer of history in a college of Kolkata for 2 years teaching students of 1st ,2nd and 3rd year. I taught Ancient and Modern Indian history there.I have taught in another school in Mohali,Punjab teaching students from classes 9-12.Presently I am working as an online tutor with concept tutors,Bangalore,India(Carve Niche Pvt.Ltd.) for the last 1year and also have been appointed as an online history tutor by Course Hero(California,U.S) and Vidyalai.com(Chennai,India).
4.00+
2+ Reviews
10+ Question Solved
Related Book For 

Question Posted:
Students also viewed these Sciences questions
-
List three specific parts of the Case Guide, Objectives and Strategy Section (See below) that you had the most difficulty understanding. Describe your current understanding of these parts. Provide...
-
1. How strong are the competitive forces confronting J. Crew in the market for specialty retail? Do a [Michael Porter] five-forces analysis to support your answer. (see chapter 3 in the textfor...
-
A portion of the housing market in New York City (and many other cities in the world) is regulated through a policy known as rent control. In essence, this policy puts a price ceiling (below the...
-
Describe the Spanish Theocratic Rule during Spain's colonial era in the Philippines?
-
Your roommate, Betty Melton, is confused about sunk costs. Explain to your roommate the meaning of sunk costs and their relevance to a decision to retain or replace equipment.
-
For each of the following items, indicate whether or not the balance per books should be adjusted. For each item that affects the balance per books, indicate whether the item should be added to (+)...
-
In spring 1989, Michael Jordan and the Chicago Bulls were in Indianapolis, Indiana, to play against the Indiana Pacers. At the same time, Karla Knafel was singing with a band at a hotel in...
-
(Entries for Bond TransactionsEffective-Interest) Assume the same information as in E14-4, except that Foreman Company uses the effective-interest method of amortization for bond premium or discount....
-
A particle undergoing simple harmonic motion has a potential energy given by E/J=3(y/m) as it oscillates back and forth along the y-axis. When the y-coordinate of the particle is 0.75 m, what is the...
-
Use the structure of the DielsAlder adduct to deduce the structure of the product X in the reaction given in Fig. P15.82. Then give a curved-arrow mechanism for the formation of X. H Cl Figure...
-
Invoking Hammonds postulate and the properties of the carbocation intermediates, explain why the doubly allylic alkyl halide A undergoes much more rapid solvolysis in aqueous acetone than compound B....
-
The heavy regions of parts typically are placed in the drag in sand casting and not in the cope. Explain why.
-
What types of regulations must businesses comply with in different jurisdictions?
-
Why is the cash budget not the same as the projected income statement? What items included on the projected income statement are not included on the cash budget?
-
What distinguishes a sole proprietorship from a company and a partnership?
-
Compute the matrix exponential e At for each system x' = Ax given in Problems 9 through 20. x' 1 = 13x 1 + 4x 2 , x' 2 = 4x 1 + 7x 2 Data in Problem 9 through 20 9. x = 5x14x2, x = 2x1-x2 x = 4x14x2...
-
Describe the factors affecting the business environment and the impact of each of these factors.
-
Laura makes the following interest-free loans during the current year. Discuss the income tax implications of each loan for both Laura and the borrower. In all cases, the applicable federal interest...
-
How will relating product contribution margin s to the amount of the constrained resource they consume help a company maximize its profits?
-
Convert the following Fischer projections into tetrahedral representations, and assign R or S stereochemistry toeach: H (b) (a) H2N- - (c) - - CH-CH CH CH
-
Which of the following Fischer projections of glyceraldehydes represent the sameenantiomer? CH- - 3 - -CH- - - CH2 A
-
Redraw the following molecule as a Fischer projection, and assign R or S configuration to the chirality center (yellow-green =Cl):
-
For the following project, the IRR and the Rate that maximizes the NPV are: Project A 0 -500 100 -8 1 28 100 38 100 48 100 100 58 6 7 8 9 100 100 100 100 10 -150
-
Define the bottleneck effect. What category of evolutionary mechanism does the bottleneck effect belong to ? Give me an example of the bottleneck effect,
-
Mrs. Johnson founded a chemical manufacturing company 25 years ago and built a successful business, which is now in a mature growth stage. She owns 100% of the company and is thinking about retiring...

Study smarter with the SolutionInn App


