Which of the following carbocations is likely to rearrange? If rearrangement occurs, give the structures of the
Question:
Which of the following carbocations is likely to rearrange? If rearrangement occurs, give the structures of the rearranged carbocations.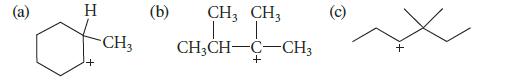
Transcribed Image Text:
@ H I + CH3 (b) CH3 CH3 T T CH3CH-C-CH3 + O
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 100% (1 review)
Rearrangement will occur when a carbocation can rearrange b...View the full answer

Answered By

Utsab mitra
I have the expertise to deliver these subjects to college and higher-level students. The services would involve only solving assignments, homework help, and others.
I have experience in delivering these subjects for the last 6 years on a freelancing basis in different companies around the globe. I am CMA certified and CGMA UK. I have professional experience of 18 years in the industry involved in the manufacturing company and IT implementation experience of over 12 years.
I have delivered this help to students effortlessly, which is essential to give the students a good grade in their studies.
3.50+
2+ Reviews
10+ Question Solved
Related Book For 

Question Posted:
Students also viewed these Sciences questions
-
Each of the following carbocations can rearrange to a more stable ion. Propose structures for the likely rearrangement products. H, (a) CH3CH2CH2CH2* (b) CH3CHCHCH3 CH CH CH2* (c)
-
The following questions are adapted from a variety of sources including questions developed by the AICPA Board of Examiners and those used in the Kaplan CPA Review Course to study property, plant,...
-
Each of the following carbocations has the potential to rearrange to a more stable one. Write the structure of the rearranged carbocation. (a) CH3CH2CH2+ (b) (c) (d) (CH3CH2)3CCH2+ (e) (CH32CHCHCHa...
-
1. Consider a local department store which only sells jeans (J) and sweaters (S). Jeans cost $20 and sweaters cost $30. For each of the examples below, graph the budget constraint our consumer faces,...
-
Why was Konop particularly eager to keep unauthorized people out of his web site?
-
The CDG Carlos, Dan, and Gail Partnership has decided to liquidate as of December 1, 20X6. A balance sheet on the date follows: Additional Information 1. Each partners personal assets (excluding...
-
McDonald Consulting, Inc., completed the following transactions during December 2010, its first month of operations: Requirements 1. Open, or set up, T-accounts in the ledger: Cash, Accounts...
-
During the school year, the Homebush School band arranges concert dates in many communities. Because only part of the school??s travel expenses are covered by the concert admission fees, the band...
-
Given triangle ABC with angle B= 85 degree, angle C = 47 degrees and a = 19 inches, what is the length of b
-
Only one of the following three alkyl halides can be prepared as the major product of the addition of HBr to an alkene. Which compound can be prepared in this way? Explain why the other two cannot be...
-
In each case, give two different alkene starting materials that would react with HBr to give the compound shown as the major (or only) addition product. (a) Br (b) Me Br
-
The starting point for computing alternative minimum taxable income is regular taxable income. What are some of the plus adjustments, plus or minus adjustments, and minus adjustments to regular...
-
Name three types of retirement plans in Puerto rico. What is a relict estate in Puerto rico? What do excise duties consist of in Puerto rico?
-
At January 1 , 2 0 2 4 , Caf Med leased restaurant equipment from Crescent Corporation under a nine - year lease agreement. The lease agreement specifies annual payments of $ 2 9 , 0 0 0 beginning...
-
what ways do enzymes contribute to metabolic pathways, and how is their activity regulated to maintain homeostasis in cells ?
-
You work in the HR team at the hospitality co-operative. The organisation has monthly mentorship sessions, but now the HR team is also planning to organise mentorship sessions for the women employees...
-
Visit the IRCC Website at https://www.canada.ca/en/immigration-refugees-citizenship.html and do your best to fill in the information below. To find the answers to most of these questions, you need to...
-
Five observations taken for two variables follow. a. Develop a scatter diagram for these data. b. What does the scatter diagram indicate about a relationship between x and y? c. Compute and interpret...
-
You continue to work in the corporate office for a nationwide convenience store franchise that operates nearly 10,000 stores. The per- store daily customer count (i.e., the mean number of customers...
-
Give the IUPAC name for the following hydrocarbon, and convert the drawing into a skeletal structure.
-
Make a graph of potential energy versus angle of bond rotation for propane, and assign values to the energy maxima.
-
Consider 2-methylpropane (isobutene). Sighting along the C2-C1 bond: (a) Draw a Newman projection of the most stable conformation. (b) Draw a Newman projection of the least stable conformation. (c)...
-
If f(x) = 3 cos(4 ln(x)), find f'(x). Answer:
-
Find an equation of the line tangent to the graph of f(x)= (3x+2) (3x-2) at the point (1,5). The equation of the line tangent to the graph of fix)=(3x+2) (3x-2) at the point (1,5) is (Type an...
-
The Student Government Association is making Mother's Day gift baskets to sell at a fund-raiser. If the SGA makes a larger quantity of baskets, it can purchase materials in bulk. The total cost (in...

Study smarter with the SolutionInn App


