Which of the molecules in each of the following pairs should have identical IR spectra, and which
Question:
Which of the molecules in each of the following pairs should have identical IR spectra, and which should have different IR spectra (if only slightly different)? Explain your reasoning carefully.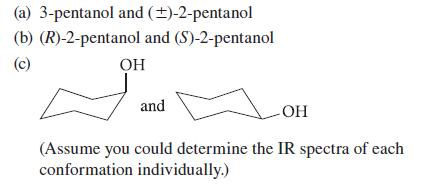
Transcribed Image Text:
(a) 3-pentanol and (±)-2-pentanol (b) (R)-2-pentanol and (S)-2-pentanol (c) OH and -OH (Assume you could determine the IR spectra of each conformation individually.)
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 50% (2 reviews)
The principle is that compounds that have different physical properties have diffe...View the full answer

Answered By

Utsab mitra
I have the expertise to deliver these subjects to college and higher-level students. The services would involve only solving assignments, homework help, and others.
I have experience in delivering these subjects for the last 6 years on a freelancing basis in different companies around the globe. I am CMA certified and CGMA UK. I have professional experience of 18 years in the industry involved in the manufacturing company and IT implementation experience of over 12 years.
I have delivered this help to students effortlessly, which is essential to give the students a good grade in their studies.
3.50+
2+ Reviews
10+ Question Solved
Related Book For 

Question Posted:
Students also viewed these Sciences questions
-
If the NMR spectra of the molecules in Problem 30 were recorded by using high resolution for each nucleus, what differences would be observed?
-
Which of the molecules in Exercise 88 have net dipole moments (are polar)? SeO3 SeO2 PCl3 SCl2 SiF4
-
Which of the molecules in Exercises 91 and 92 have net dipole moments (are polar)? Exercise 91. a. XeCl2 b. ICl3 c. TeF4 d. PCl5 Exercise 92. a. ICl5 b. XeCl4 c. SeCl6
-
Net income Depreciation expense Accounts receivable increase (decrease) Inventory increase (decrease) Accounts payable increase (decrease) Accrued liabilities increase (decrease) O Changes in current...
-
What are the purpose and importance of a procedures manual?
-
Fair-to-Midland Manufacturing, Inc., (FMM) has applied for a loan at True Credit Bank. Jon Fulkerson, the credit analyst at the bank, has gathered the following information from the companys...
-
The Anchor Glass Container Corporation and its parent company, Consumers Packaging, Inc. (CPI), entered into a series of agreements with Encore Glass, Inc., to supply glass containers of a specific...
-
Eastbay Hospital has an auxiliary generator that is used when power failures occur. The generator is worn out and must be either overhauled or replaced with a new generator. The hospital has...
-
Unpolarized sunlight goes through two pairs of sunglasses. The second pair is oriented 20 o respect to the first. What is the exiting intensity in terms of the initial intensity I0?
-
List the factors that determine the wavenumber of an infrared absorption.
-
The mass spectrum of 2-chloropentane shows large and almost equally intense peaks at m/z = 71 and m/z = 70. (a) Classify each peak as an even-electron or odd-electron ion. (b) What stable neutral...
-
If f(x) = [x] + [-x], show that limx2 f(x) exists but is not equal to f(2).
-
Check your own state statute for its version of the UCC.
-
For each of the following studies, decide whether to illustrate the described outcomes with a line graph or a bar graph; then create graphs that accurately portray the outcomes. 1. In a study of the...
-
Which of the following traversals always gives the sorted sequence of the elements in a BST? Preorder Ignored Postorder Undefined
-
Explain the functions of a clearinghouse associated with a futures exchange.
-
How do margin requirements in the futures market differ from margin requirements in the cash market?
-
Lorene, Inc., owns an apartment complex. The terms of Lorenes lease agreement require new tenants to pay the first and last months rent and a cleaning deposit at the inception of the lease. The...
-
The comparative statements of financial position of Menachem NV at the beginning and end of the year 2019 appear below. Net income of ¬34,000 was reported, and dividends of ¬23,000 were paid...
-
Without looking at Table 24.2, rank the following compounds in order of ascending basicity. (a) p-Nitro aniline, p-aminobenzaldehyde, p-Bromoaniline (b) p-Chloroanilinc, p-aminoacetophenone, p-methyl...
-
Calculate the percentages of neutral and protonated forms present in a solution of 0.0010 M pyrimidine at pH = 7.3. The pKa of pyrimidinium ion is 1.3.
-
Propose structures for either a nitrile or an amide that might be a precursor of each of the following amines: (a) CH3CH2CH2NH2 (b) (CH3CH2CH2)2NH (c) Benzyl amine, C6H5C2NH2 (d) N-Ethyl aniline
-
Regency Integrated Chips (RIC), a large Nashville-based technology company is evaluating a new project to manufacture a new chip. a. The project's estimated economic life is 5 years. b. RIC's...
-
For a boat to float in a tidal bay, the water must be at least 2.7 meters deep. The depth of the water around the boat, d (t), in meters, where t is measured in hours since midnight, is d(t)=5+4.6...
-
When journalizing an unrealized holding loss on FV-OCI debt securities, how and where should a company record the loss? Explain.

Study smarter with the SolutionInn App


