Which of the two phenols in each set is more acidic? Explain. (a) 2,5-dinitrophenol or 2,4-dinitrophenol (b)
Question:
Which of the two phenols in each set is more acidic? Explain.
(a) 2,5-dinitrophenol or 2,4-dinitrophenol
(b) phenol or m-chlorophenol
(c)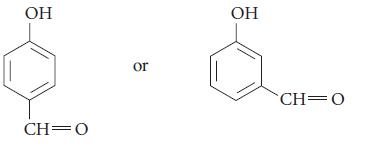
Transcribed Image Text:
OH CH 0 or OH CH=0
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 100% (1 review)
a 24Dinitrophenol is more acidic because its conjugatebase anion has more important re...View the full answer

Answered By

Nyron Beeput
I am an active educator and professional tutor with substantial experience in Biology and General Science. The past two years I have been tutoring online intensively with high school and college students. I have been teaching for four years and this experience has helped me to hone skills such as patience, dedication and flexibility. I work at the pace of my students and ensure that they understand.
My method of using real life examples that my students can relate to has helped them grasp concepts more readily. I also help students learn how to apply their knowledge and they appreciate that very much.
4.00+
1+ Reviews
10+ Question Solved
Related Book For 

Question Posted:
Students also viewed these Sciences questions
-
Which of the, two phenols in each set is more acidic? Explain. Phenol or m-chlorophenol
-
Maya earns $50/hour. John earns $45/hour + 5% commission + bonus (commission and bonus are based on his performance). (Note: Some economics concepts you may consider -- opportunity cost, law of...
-
The cash records and bank statement for the month of July for Ravine Incorporated are shown below. RAVINE INCORPORATED Cash Account Records July 1 , 2 0 2 4 , to July 3 1 , 2 0 2 4 Cash Balance July...
-
Assume electricity is generated by three technologies: a base load technology with cost function C b (x b ) and proportional emission coefficient b ; gas turbines with cost function C g (x g ) and...
-
Show that the clock in S reads 100 min when the light flash reaches B, which is traveling away from C with speed 0.6c. y:V= 0.6c -100 cmin- A' Flashbulb
-
Imagine that you are trying to evaluate the economics of purchasing an automobile. You expect the car to provide annual after-tax cash benefits of $1,200 at the end of each year and assume that you...
-
Briefly explain the dual-kernel approach to RTOS design.
-
Max Small has outstanding school loans that require a monthly payment of $1,000. He needs to purchase a new car for work and estimates that this will add $350 per month to his existing monthly...
-
If you were a stake holder or someone that is reviewing a financial report and you see a change is made to one of the methods that is always used and it is bringing some inaccurate results, how would...
-
The following compound, unlike most phenols, is soluble in neutral aqueous solution, but insoluble in aqueous base. Explain this unusual behavior. H -N(CH3)3 CI
-
Suggest a mechanism for the oxo reaction (Eq. 18.63) involving intermediates that are consistent with the 16- and 18-electron rules. H,C=CH2 + H2 + CO ethylene HCO (CO)4 100-120 C CHCHCH...
-
Indicate in Fig. the region in which each of the figures should be placed. Figures that measure less than -in 1 / Blue figures across IV v/ VI VII Circles VIII
-
Explain the following terms: (a) Grant date (b) Measurement date (c) Vesting date (d) Vesting conditions (e) Forfeiture rate
-
The consequences of climate change on the economy is a popular topic in the media. Suppose that a series of wildfires destroys crops in the western states at the same time a hurricane destroys...
-
In what situations can a hedged item be either a fair value hedge or a cash flow hedge? Explain why each type of hedge is applicable to the situation.
-
A firm that intends to hedge against commodity price risk has to decide whether to use a forward contract or an options contract to hedge the risk. What factors should the firm consider before...
-
Company X purchased $2 million face value of bonds issued by company Y on 2 January 2013 at purchase cost of $1.94 million. The bond had coupon of 5% payable annually, and matures on 31 December...
-
An office in Albuquerque has 24 workers including management. Eight of the workers commute to work from the west side of the Rio Grande River. Suppose six of the office workers are randomly selected....
-
Question 6.10 Current and deferred tax worksheets and tax entries From the hip Ltd?s statement of profit or loss for the year ended 30 June 2007 and extracts from its statements of financial position...
-
The following names are all incorrect or incomplete, but they represent real structures. Draw each structure and name it correctly. (a) 2-ethylpentane (b) 3-isopropylhexane (c)...
-
Provide IUPAC names for the following compounds. (a) (CH3)2CHCH2CH3 (b) CH3-C(CH3)2-CH3 (c) (d) (e) (f) CH CH CHCH CH,CHCHCH le ' CH CH,CH, CH CH CH, CH CH C(CH CH,CH,CHCHCH, CH(CH2 CH CHCH,CH, CH)C...
-
In each pair of compounds, which compound has the higher boiling point? Explain your reasoning. (a) octane or 2,2,3-trimethylpentane (b) Nonane or 2-methylheptane (c) 2, 2, 5-trimethylhexane or nonane
-
Discuss how you and other employees use both commonsense theory and working theory at your current or previous workplace. Be sure to explain your points using specific examples.
-
TO Points Calgary Corporation has PEs in Alberta, British Columbia and in the United States. Gross revenues, Net Income and salaries and wages for each PE are: Alberta British Columbia United States...
-
Apparent and actual authority are concepts applied in what areas of law? explain

Study smarter with the SolutionInn App


