Ascorbic acid (vitamin C) does not contain a traditional carboxylic acid group, but it is, nevertheless, still
Question:
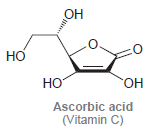
Он но но ОН Ascorbic acid (Vitamin C)
Step by Step Answer:
The proton highlighted above is the most acidic ...View the full answer



Related Video
Lemon juice preserves apples by slowing down the oxidation process. Oxidation is a chemical reaction that occurs when oxygen reacts with certain substances, such as apples. When an apple is cut or bitten, oxygen is exposed to the inside of the apple and causes enzymes in the apple to turn brown, which is an indication of oxidation. The browning process is caused by the production of polyphenol oxidase (PPO) enzymes that convert phenolic compounds into quinones, which then polymerize to form the brown pigments. One of the compounds present in lemon juice is ascorbic acid (vitamin C), which is a natural antioxidant. Antioxidants work by neutralizing the free radicals that cause oxidation. When lemon juice is applied to apples, the ascorbic acid in the lemon juice reacts with the PPO enzymes and slows down the browning process. You can do an experiment by cutting apples into small pieces, leaving one apple piece in contact with air and the others covered with lemon juice and compare the browning process. This will help to understand the antioxidation process in fruits.
Students also viewed these Sciences questions
-
Does ascorbic acid (vitamin C) have a D or Lconfiguration? HO . C=0 Ascorbic acid . -H - CH2OH
-
Ascorbic acid (vitamin C, C6H8O6) is a water-soluble vitamin.
-
Suggest reasonable explanations for each of the following observations. (a) Both hydrogens are anti to each other in the most stable conformation of formic acid. (b) Oxalic acid has a dipole moment...
-
In Exercises, find the limit. lim 5 x 0-x 813 X
-
Lynne Catering uses two measures of activity, jobs and meals, in the cost formulas in its budgets and performance reports. The cost formula for catering supplies is $480 per month plus $91 per job...
-
Show that a 50-J change in kinetic energy occurs when a model airplane on takeoff is moved a distance of 5 m by a sustained net force of 10 N.
-
Class Year by Gender Exercise 2.35 deals with the distribution of class Year for students in an introductory statistics course. The StudentSurvey data also has information on the Gender for each...
-
Return to the case of the diagnostic scanner discussed in Problems 1 through 6. Suppose the entire $5,200,000 purchase price of the scanner is borrowed. The rate on the loan is 8 percent, and the...
-
Admitting New Partners Who Buy an Interest and Contribute Assets The capital accounts of Trent Henry and Tim Chou have balances of $117,500 and $84,800, respectively. LeAnne Gilbert and Becky Clarke...
-
Over a 14-day period, data were collected on the number of passengers travelling on two ferries, A and Z. The results are presented to the right. a. How many more passengers travelled on ferry Z than...
-
In each compound below, two protons are clearly identified. Determine which of the two protons is more acidic. (a) (b) (c) (d) (e) (f) -N
-
In the following compound two protons are clearly identified. Determine which of the two is more acidic. After comparing the conjugate bases, you should get stuck on the following question: Is it...
-
In the Val IT framework, portfolio management includes which of the following? a. Establishing strategic priorities b. Determining required characteristics of the portfolio c. Monitoring performance...
-
How your MUTED GROUP THEORY is demonstrated by the movie Legally Blonde . Make a thesis statement based on that premise.
-
The investor buys 100 shares of stock for $10 a share and sells it for $14. Also assume the margin requirement is 60%, the commission rate is 5% of the purchase or sale price, the interest rate is...
-
Discuss the concept of popular culture and explain how popular culture is related with mass media.? Give some examples of popular culture and elaborate relationship between popular culture and...
-
Describe a few differences between children's temperaments in different cultures and why those temperaments might benefit them in their environments. What factors influence?
-
Suppose you have been assigned an important position in Foreign Office where meeting people from different backgrounds and culture is normal practice. How would you transform your communication...
-
Name the quadrant in which the angle lies. csc > 0, cos < 0
-
Pedro Bourbone is the founder and owner of a highly successful small business and, over the past several years, has accumulated a significant amount of personal wealth. His portfolio of stocks and...
-
Chlorination reactions of certain alkanes can be used for laboratory preparations. Examples are the preparation of chlorocyclopropane from cyclopropane and chlorocyclobutane from cyclobutane. What...
-
Each of the following alkanes reacts with chlorine to give a single monochloro substitution product. On the basis of this information, deduce the structure of each alkane. (a) C5H10 (b) C8H18
-
Consider the chlorination of (S)-2-chloropentane at C4. (a) Write structural formulas for the products, showing three dimensions at all chirality centers. Give each its proper (R, S) designation. (b)...
-
1. Solve FV = PMT((1+r/m)mt 1) for PMT. PMT = r/m 2. Given the formula in the format to input into the calculator. PMT =
-
Bramble Enterprises purchased a machine on January 1 , 2 0 2 4 , for $ 2 2 1 0 0 . The machine had an estimated useful life of 1 0 years and an estimated residual value of $ 2 7 0 0 . Assuming...
-
For an ideal gas, what is the pressure in atm if the volume of the container is 5000 mL, the mol quantity is 0.812 mol, and the temperature is 28 degrees Celsius? PV = nRT (qu - A) (BA/MA + d) = nRT

Study smarter with the SolutionInn App


