At high temperatures, alkanes can undergo dehydrogenation to produce alkenes. For example: This reaction is used industrially
Question:
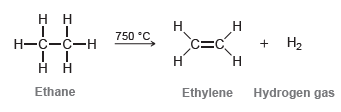
This reaction is used industrially to prepare ethylene while simultaneously serving as a source of hydrogen gas. Explain why dehydrogenation only works at high temperatures.
Transcribed Image Text:
Н. нн 750 °C. Н Н—с—с—н c=C + H2 Н Ethane Ethylene Hydrogen gas
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 64% (14 reviews)
A reaction is only favorable if G is negative Recall that G has two components H and TS ...View the full answer

Answered By

Charles mwangi
I am a postgraduate in chemistry (Industrial chemistry with management),with writing experience for more than 3 years.I have specialized in content development,questions,term papers and assignments.Majoring in chemistry,information science,management,human resource management,accounting,business law,marketing,psychology,excl expert ,education and engineering.I have tutored in other different platforms where my DNA includes three key aspects i.e,quality papers,timely and free from any academic malpractices.I frequently engage clients in each and every step to ensure quality service delivery.This is to ensure sustainability of the tutoring aspects as well as the credibility of the platform.
4.30+
2+ Reviews
10+ Question Solved
Related Book For 

Question Posted:
Students also viewed these Sciences questions
-
At high temperatures, a dynamic equilibrium exists between carbon monoxide, carbon dioxide, and solid carbon. At 850oC, Kc is 0.153. a. What is the value of Kp? b. If the original reaction system...
-
At high temperatures, a dynamic equilibrium exists between carbon monoxide, carbon dioxide, and solid carbon. At 900oC, Kc is 0.238. a. What is the value of Kp? b. Some carbon dioxide is added to the...
-
Reverse Diels-Alder reactions can occur at high temperatures. Why are high temperatures required?
-
Tonight at 7pm a professional basketball game featuring the Golden State Warriors and the Washington Wizards will be played at the Chase Center, an indoor arena in the Mission Bay neighborhood of San...
-
Create a fictional company and present a marketing campaign to brand that company. Make sure that you give me some background as to what the company sells and who you are trying to reach; your...
-
In Becker and Murphys rational addicts model, smokers are perfectly aware of the potential for smoking to cause addiction, and they take this into account when deciding whether or not to smoke....
-
With reference to the preceding exercise, find the corresponding distribution function, and use it to determine the probabilities that a random variable having the distribution function will take on...
-
Identifying cost behavior Required Identify the following costs as fixed or variable. Costs related to plane trips between San Diego, California, and Orlando, Florida, follow. Pilots are paid on a...
-
Gravel is being dumped from a conveyor belt at a rate of 10 cubic feet per minute. It forms a pile in the shape of a right circular cone whose base diameter and height are always the same. How fast...
-
Brian Fry Products manufactures a variety of machine tools and parts used primarily in industrial tasks. To control production, the company requires the information listed below. Design an efficient...
-
Identify the reagents you would use to convert 1-pentene into a geminal dibromide (geminal indicates that both bromine atoms are connected to the same carbon atom).
-
Identify the reagents you would use to convert methylcyclohexane into each of the following: (a) A 3 alkyl halide (b) A trisubstituted alkene (c) A 2 alcohol (d) 3-methylcyclohexene
-
The given numbers express angle measure. Express the measure of each angle in terms of degrees. 97 2 4 15
-
Why is the concept of probability important to understanding statistics?
-
Suppose there are four events A, B, C, and D. The following information is given: P(A) = .5 P(B) = .15 P(C) = .20 P(AUD) = .72 P(A | B) = .25 P(AC) = .04 P(AD) = .03
-
An automobile manufacturer produces cars in four different colors and offers three different options packages. How many different combinations of color and options package can the auto manufacturer...
-
Suppose you flip a fair coin once and roll a 6-sided die once. What is the probability of tossing a tail and rolling a one?
-
You believe you have come up with a fool-proof way to win at roulette. Because the odds are nearly 5050 that red will come up and nearly 5050 that black will come up in roulette, you believe that...
-
Graph several functions that satisfy the following differential equations. Then find and graph the particular function that satisfies the given initial condition. f'(x) = 2 cos 2x; f(0) = 1
-
Which of the following raises the credibility of areport? Which of the following raises the credibility of a report? Multiple Choice avoiding predictions avoiding the use of cause-effect statements...
-
How much is the reaction rate for these reactions increased or decreased if the concentration of hydroxide ion is doubled? If the concentrations of the both the alkyl chloride and hydroxide ion...
-
Show how these compounds could be prepared from alkylhalides: SCH, OCCH3 b) OCH3 a) c) Two methods Two methods
-
Show how these products could be synthesized from the indicated starting material. More than one step may be necessary. Make sure that the product has the stereo chemistryshown. Starting material...
-
Should an MNC purchase Nestl stock yes or no? Recommendations should include the company's sustainability plans, the image of the company, Gordon Growth result, and financial performance. Any...
-
Can the sponsor, or general partner, continue to achieve such high returns? Framed another way, can the general partner earn a 25+% return on the $6,305,000 received at the end of the 10th year? If...
-
Select a hospital that publicly displays their financial performance on their website. Describe the Profit Loss Statement: Review top revenue items Review cost comparison compared to previous year...

Study smarter with the SolutionInn App


