Below is the structure of rilpivirine, a promising new anti-HIV drug that combats resistant strains of HIV.
Question:
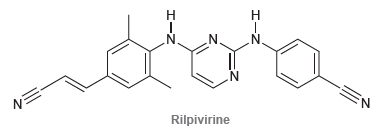
(a) Identify the two most acidic protons in rilpivirine.
(b) Identify which of these two protons is more acidic. Explain your choice.
Transcribed Image Text:
Н Н N. Rilpivirine エーZ
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 91% (12 reviews)
a The two most acidic protons are labeled H a and H b b Ha is expected to be slightly more a...View the full answer

Answered By

Bhartendu Goyal
Professional, Experienced, and Expert tutor who will provide speedy and to-the-point solutions. I have been teaching students for 5 years now in different subjects and it's truly been one of the most rewarding experiences of my life. I have also done one-to-one tutoring with 100+ students and help them achieve great subject knowledge. I have expertise in computer subjects like C++, C, Java, and Python programming and other computer Science related fields. Many of my student's parents message me that your lessons improved their children's grades and this is the best only thing you want as a tea...
3.00+
2+ Reviews
10+ Question Solved
Related Book For 

Question Posted:
Students also viewed these Sciences questions
-
Below is the structure of caffeine, but its lone pairs are not shown. Identify the location of all lone pairs in this compound: - -N N. .C. Caffeine Z-O- Z-U
-
Identify which of the following substrates will undergo an S N 1 reaction more rapidly. Explain your choice. Br Br or
-
Below (Figure 12.20) are the 1H NMR, 13C NMR, IR, and mass spectra of the compound C8H8O. What is the structure of the compound?
-
T-bills currently yield 3.9 percent. Stock in Nina Manufacturing is currently selling for $63 per share. There is no possibility that the stock will be worth less than $61 per share in one year. a....
-
When should a country attempt to depreciate its currency? How could it be done?
-
Three basketball players compete in a basketball foul shooting contest. The eligible players are allowed one foul shot per round. After each round, all players who miss their foul shots are...
-
There is some evidence that "young blood" might improve the health, both physically and cognitively, of elderly people (or mice). Exercise 2.69 on page 75 introduces one study in which old mice were...
-
Stiever Corp. has collected the following data concerning its maintenance costs for the past 6 months. Compute the variable- and fixed-cost elements using the high-lowmethod. July August September...
-
What measures can be implemented to address the growing cybersecurity threats targeting supply chain infrastructure, safeguarding critical assets and sensitive information against data breaches and...
-
Calculate the unit cell edge length for an 85 wt% Fe-15 wt% V alloy. All of the vanadium is in solid solution, and, at room temperature the crystal structure for this alloy is BCC.
-
Identify the number of sp 3 -hybridized carbon atoms in the following compound: (CH 3 ) 2 C = CHC(CH 3 ) 3
-
Propose a mechanism for the following transformation:
-
What is required to transform a business-level strategy from an idea into reality?
-
Cost of Capital The main goal of this case is to reinforce your understanding of how to estimate the cost of capital in capital budgeting. Once you finish learning Lecture 10 - Estimating the Cost of...
-
NOPAT Component Year 0 Change in Working Capital Component Year... NOPAT Component Year 0 Change in Working Capital Component Year -1 Year 0 Net CapEx Component Year 0 EBIT Accounts Receivable ...
-
Please fill out Assumptions with Formulas and Answers. Assumptions Historicals Stage 1 Stage 2 Assumption 2017 2018 2019 Average 2020 2021 2022 2023 2024 2025 2026 2027 2028...
-
Use the fund fact sheets provided by the respective asset management companies (attached) for the following mutual funds to answer questions 1-6: TD Monthly Income Fund (TDB622) (I) Investor's...
-
Identify and discuss why employees join unions. What do you believe are the major pros and cons of unions today? If you had a choice, would you recommend union formation within your organization? Why
-
A mean average of 60 on seven exams is needed to pass a course. On her first six exams, Sheryl received grades of 52, 72, 80, 65, 57, and 69. (a) What grade must she receive on her last exam to pass...
-
Write a function that reads a Float24_t value: Float24_t float24_read(void) A legitimate float24 value string is of the form: "mantissabexponent" where the mantissa (m) and the exponent (e) may have...
-
Dehydration of trans-2-methylcyclopentanol with POCl3 in pyridine yields predominantly 3-methylcyclopentene. Is the stereochemistry of this dehydration syn or anti? Can you suggest a reason for...
-
How would you synthesize the following alcohols, starting with benzene and other alcohols of six or fewer carbons as your only organic reagents? CH3 (a) (b) CH3CH2CH2HCH2CH2OH "CH-CH (c) (d) CH ...
-
2, 3-Dimethyl-2, 3-butanediol has the common name pinacol. On heating with aqueous add, pinacol rearranges to pinacolone, 3, 3-dimethyl-2-hutanone. Suggest a mechanism for this reaction. CH :-C CH ...
-
PART A (Retained Earnings Statement) January 1, Retained Earnings Beginning Balance 2021 Income before income tax 1 $2,340,000 Prior period adjustment: understatement of 2020 depreciation expense...
-
There are 3 source and 4 destinations, For the data below, construct an initial feasible solution. Cost Destination 1 Destination 2 Destination 3 Destination 4 Supply Source 1 12 18 9 11 105 Source 2...
-
A company is using Dynamics 3 6 5 Project Operations. You acquire an independent software vendor ( ISV ) solution from AppSource that contains several project charts and install the solution in the...

Study smarter with the SolutionInn App


