Compare the following two constitutional isomers. The 13 C NMR spectrum of the first compound exhibits five
Question:
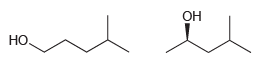
Transcribed Image Text:
Он НО.
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 80% (15 reviews)
The first compound lacks a chirality center The two methyl groups are enantiot...View the full answer

Answered By

Nazrin Ziad
I am a post graduate in Zoology with specialization in Entomology.I also have a Bachelor degree in Education.I posess more than 10 years of teaching as well as tutoring experience.I have done a project on histopathological analysis on alcohol treated liver of Albino Mice.
I can deal with every field under Biology from basic to advanced level.I can also guide you for your project works related to biological subjects other than tutoring.You can also seek my help for cracking competitive exams with biology as one of the subjects.
3.30+
2+ Reviews
10+ Question Solved
Related Book For 

Question Posted:
Students also viewed these Sciences questions
-
Draw two constitutional isomers of cis-1, 2-dibromo-cyclopentane.
-
Write structural formulas of the type indicated: (a) Bond-line formulas for seven constitutional isomers with the formula C4H10O; (b) Condensed structural formulas for two constitutional isomers with...
-
Two constitutional isomers having molecular formula C4H6O are both symmetrical in structure. In their infrared spectra, neither isomer when in dilute solution in CCl4 (used because it is nonpolar)...
-
An employee of Gus's Restaurant sustains a work-related injury. The owner of the restaurant is liable for the injury, however, the employee is not covered under the state's workers' compensation...
-
What are the differences between a managerial and a technical designer of a DSS?
-
What other deductions might be made from gross pay by an employer?
-
Estimate the model parameters for the gasoline mileage data using principalcomponent regression. a. How much has the residual sum of squares increased compared to least squares? b. How much shrinkage...
-
Ebersoll Mining has $6 million in sales, its ROE is 12%, and its total assets turnover is 3.2. The company is 50% equity financed. What is its net income?
-
5. In an experiment, we roll a dice twice and document X as the result of first roll and X2 as the result of the second roll: a. We are interested in the outcome as the sequence of two rolls, i.e.,...
-
Identify at least three noneconomic attributes that may be used as evaluation criteria in the decision making process.
-
Explain why the effective interest rate (r EAR ) on a loan often is much higher than the simple, or stated, interest rate (APR).
-
Susan Visscher, owner of Visschers Hardware, is negotiating with First Merchants Bank for a $50,000, one-year loan. First Merchants has offered Visscher the following alternatives. Calculate the...
-
Melissa (who is not a Scottish taxpayer) owns a house which she lets to tenants. The house was let throughout 2021-22 and rents received during the year were 7,300. Her deductible expenditure for the...
-
121. In genetic engineering, a DNA segment (gene) of interest, is transferred to the host cell through a vector. Consider the following four agents (A-D) in this regard and select the correct option...
-
133. In chasmogamy pollination takes place in (1) open flower (3) large flower (2) closed flower (4) geitonogamy flower 134. Identify the type of flower A and B. (1) A-Cleistogamous; B-Chasmogamous...
-
140. Which of the following molecules are never found as polymers? (1) Carbohydrates (2) Lipids (3) Nucleic Acids (4) Proteins
-
136. To cleave pBR322 at the rop site which restriction enzyme is used? (1) PvuI (3) Pst II (2) Pvu II (4) Hind III 137. Biolistics is a method of? (1) Artificial gene synthesis (2) Identification of...
-
129. Many ovules are present in the ovary of: (1) Wheat (3) Papaya (2) Rice (Paddy) (4) Mango 130. Which hydrophytic plant is not pollinated by water? (1) Vallisneria (3) Water hyacinth (2) Hydrilla...
-
(a) What is a population? (b) What is a sample?
-
Suppose the market is semistrong form efficient. Can you expect to earn excess returns if you make trades based on? a. Your brokers information about record earnings for a stock? b. Rumors about a...
-
The amino acid methionine is biosynthesized by a multistep route that includes reaction of an imine of pyridoxal phosphate (PLP) to give an unsaturated imine, which then reacts with cysteine. What...
-
Each of the following reaction schemes contains one or more flaws. What is wrong in each case? How would you correct eachscheme? (a) Ag*, NH,OH 1. CH3MgBr 2. * CH , I b) H H2 CH * CHgCHHIO)2 (c) ...
-
6-Methyl-5-hepten-2-one is a constituent of lemongrass oil. How could you synthesize this substance from methyl4-oxopentanoate? CH3CH2CH2OCH3 Methyl 4-oxopentanoate
-
1. How does marketing create value, and how do firms become more value driven? 2. Describe a firm's stakeholders and how they are impacted by conscious marketing.
-
Beginning inventory 100 units @ $8.00 = $ 800 Purchase # 1 200 units @ $6.00 = 1,200 Purchase # 2 100 units @ $12.00 = 1,200 Total 400 units $3,200 Ending inventory is 150 units. 1. What is cost of...
-
Sam's Ice Creams, a cottage country favourite in Northern Ontario, has launched a new range of ice creams branded as "Summer Flavours." To capitalize on the cottage and tourism season from May to...

Study smarter with the SolutionInn App


