Consider the following reaction. Predict whether an increase in temperature will favor reactants or products. Justify your
Question:
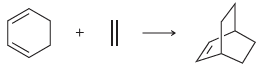
Transcribed Image Text:
+
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 87% (16 reviews)
Recall that G has two components H and TS We must analyze each term separately The first term is exp...View the full answer

Answered By

Cyrus Sandoval
I a web and systems developer with a vast array of knowledge in many different front end and back end languages, responsive frameworks, databases, and best code practices. My objective is simply to be the best web developer that i can be and to contribute to the technology industry all that i know and i can do. My skills include:
- Front end languages: css, HTML, Javascript, XML
- Frameworks: Angular, Jquery, Bootstrap, Jasmine, Mocha
- Back End Languages: Java, Javascript, PHP,kotlin
- Databases: MySQL, PostegreSQL, Mongo, Cassandra
- Tools: Atom, Aptana, Eclipse, Android Studio, Notepad++, Netbeans.
Having a degree in Computer Science enabled me to deeply learn most of the things regarding programming, and i believe that my understanding of problem solving and complex algorithms are also skills that have and will continue to contribute to my overall success as a developer.
I’ve worked on countless freelance projects and have been involved with a handful of notable startups. Also while freelancing I was involved in doing other IT tasks requiring the use of computers from working with data, content creation and transcription.
5.00+
1+ Reviews
10+ Question Solved
Related Book For 

Question Posted:
Students also viewed these Sciences questions
-
Consider the following reaction to produce methyl acetate: When this reaction is carried out with CH3OH containing radioactive oxygen-18, the water produced is not radioactive. Explain. CH,OH CH...
-
Consider the following reaction at some temperature: H2O(g) + CO(g) H2(g) + CO2(g) K = 2.0 Some molecules of H2O and CO are placed in a 1.0- L container as shown below. When equilibrium is reached,...
-
Consider the following reaction at 248oC and 1.00 atm: CH3Cl(g) + H2(g) CH4(g) + HCl(g) For this reaction, the enthalpy change at 248oC is 283.3 kJ/ mol. At constant pressure the molar heat...
-
Suppose that one worker can shovel snow from a storefront sidewalk in 50 minutes and another worker can shovel it in 30 minutes. How long will it take if they work together?
-
1. Develop and submit the Mini-Case Just Right Globalization 2. Is there more to the "not-invented-here" syndrome than simply hurt feelings on the part of those who believe they are being dictated to...
-
Refer to Exercise 56. Suppose 9 of the 13 stocks are growth stocks and four are income stocks. In Exercise 56 Samir is selecting stocks for his portfolio. He has 13 candidate stocks from which he...
-
An orthotropic lamina has thermal expansion coefficients \(\alpha_{1}=-4.0\) \(\left(10^{-6} ight) \mathrm{m} / \mathrm{m} / \mathrm{K}\) and \(\alpha_{2}=79\left(10^{-6} ight) \mathrm{m} /...
-
The following events apply to The Ice Cream Parlor for the 2013 fiscal year: 1. The company started when it acquired $20,000 cash from the issue of common stock. 2. Purchased a new ice cream machine...
-
Watch the video of the Frontline documentary about questionable police shootings by the New Orleans Police Department in the wake of Hurricane Katrina. Develop an essay that discusses the possible...
-
Graphically verify Eq. (1-13) for a lead absorber and Mo Ka, Rh Ka, and Ag Ka radiation. (The mass absorption coefficients of lead for these radiations are 122.8, 84.13, and 66.14 cm/g,...
-
In the presence of a special type of catalyst, hydrogen gas will add across a triple bond to produce a double bond: The process is exothermic. Do you expect a high temperature to favor products or...
-
When an amine is protonated, the resulting ammonium ion is not electrophilic: However, when an imine is protonated, the resulting iminium ion is highly electrophilic: Explain this difference in...
-
Find the absolute maximum and absolute minimum values of f on the given interval. (a) f(x) = 12 + 4x - x2, [0, 5] (b) f(x) = 2x3 - 3x2 - 12x + 1, [-2, 3] (c) f(x) = x3 - 6x2 + 5, [-3, 5]
-
Why is the cost allocation process important to health services managers?
-
It might be counter-intuitive, but knowing when to walk away is sometimes the best course of action. When is it a salespersons best course of action?
-
Describe the three forms of expansion selling.
-
How can income statement data be used to estimate cash flow?
-
In the Ricardian trade model, comparative advantage is determined by: A. technology. B. the capital-to-labor ratio. C. the level of labor productivity.
-
Suppose that a consumer always consumes 2 spoons of sugar with each cup of coffee. If the price of sugar is p 1 per spoonful and the price of coffee is p 2 per cup and the consumer has m dollars to...
-
A certain Christmas tree ornament is a silver sphere having a diameter of 8.50 cm. Determine an object location for which the size of the reflected image is three-fourths the size of the object. Use...
-
If we examine Table 21.1, we find that the methylphenols (cresols) are less acidic than phenol itself. For example, This behavior is characteristic of phenols bearing electron-releasing groups....
-
When o-chlorotoluene is subjected to the conditions used in the Dow process (i.e., aqueous NaOH at 350oC at high pressure), the products of the reaction are o-cresol and m-cresol. What does this...
-
When 2-bromo-1, 3-dimethylbenzene is treated with sodium amide in liquid ammonia, no substitution takes place. This result can be interpreted as providing evidence for the elimination-addition...
-
Electric energy units are often expressed in "kilowatt-hours." (a) Show that one kilowatt-hour (k Wh) is equal to 3.6 x 10 J. (b) If a typical family of four uses electric energy at an average rate...
-
What are the ethical and safety implications of deadlocks in critical infrastructure systems, such as power grids or medical devices, and what measures can be taken to ensure high reliability and...
-
Discuss the economic rationale for fiscal decentralisation with reference to the Tiebout model. How would you characterise the empirical support for the model's implications?

Study smarter with the SolutionInn App


